
Analyze the FDA AI early-phase clinical trials RFI. This guide covers pilot program objectives, AI evaluation metrics, and evidence-based response strategies.

960 articles

Analyze the FDA AI early-phase clinical trials RFI. This guide covers pilot program objectives, AI evaluation metrics, and evidence-based response strategies.

Review this educational playbook for Veeva CRM Approved Email implementation. Learn about technical setup, compliance frameworks, HCP engagement, and pitfalls.

Examine Phase 2/3 trial results for TAK-881, a 20% SCIG therapy for primary immunodeficiency offering comparable IgG exposure in half the infusion volume.

Examine AI in pharma HEOR and real-world evidence. This report details how machine learning and RWD advance health economic modeling and regulatory decisions.

Review the FDA full approval of Tecartus for relapsed mantle cell lymphoma. This analysis covers ZUMA-2 Cohort 3 efficacy data, CAR-T safety, and clinical use.

Review the Veeva Vault 26R2 release preview. Learn about mid-2026 platform enhancements, including document viewer upgrades, QMS, RIM, and safety workflows.

Analyze the 2026 biotech capital outlook, detailing Blackstone's record $6.3B Life Sciences Fund VI, VC funding trends, biopharma M&A, and market recovery.

Review the 2026 pharma AI vendor landscape. This analysis details 150+ companies using machine learning for drug discovery, clinical trials, and manufacturing.

Analyze the BeOne-Huahui licensing agreement for the HH160 PD-1/CTLA-4/VEGF trispecific antibody. Review recent China biotech cross-border licensing trends.

Examine the FDA approval of Otarmeni, the first gene therapy for OTOF-related congenital hearing loss, and its expedited National Priority Voucher review.

Analyze AcuityMD's $80M Series C funding and the role of AcuityAI. Learn how agentic AI optimizes MedTech commercial operations, sales, and market access.

Analyze how AI clinical trial recruitment addresses enrollment bottlenecks. This report examines Iterative Health's $77M expansion into cardiology and obesity.

Examine the $2.25B Eli Lilly-Profluent deal. Learn how generative AI and protein design are creating novel recombinases for kilobase-scale genome editing.

A 2026 comparison guide of open-source LIMS platforms for life science labs. Learn about system architectures, customizability, and compliance trade-offs.

Examine the 2026 FDA real-time clinical trial pilot. This report analyzes how AI and cloud platforms enable continuous oncology data monitoring for new drugs.

Understand why large language models (LLMs) improve performance when given high-stakes or emotional prompts, and explore research on the EmotionPrompt effect.

Analyze LLM position bias, including primacy and recency effects in transformer models. Learn how prompt structure impacts attention and generation accuracy.

Examine the FDA's first warning letter for AI misuse in cGMP manufacturing. This review details compliance lessons and human oversight requirements for QA.

Examine the impact of Novartis CEO Vas Narasimhan joining Anthropic's board. This report analyzes pharma AI governance, healthcare regulations, and drug R&D.

Review this 2026 vendor comparison of clinical trial payment software. Analyze automated site payment and participant stipend management system capabilities.
Analyze Q1 2026 biopharma M&A trends, including 19 billion-dollar deals. Learn how patent cliffs and AI valuation drivers impact pharmaceutical acquisitions.

Analyze the role of multi-agent AI and co-scientist architectures, including withZeta.ai, in accelerating rare cancer drug discovery and modern oncology R&D.

Review AI molecule prioritization methods and computational triage tools used in drug discovery pipelines to filter generative AI candidates via ADMET and QSAR.

Read a comprehensive analysis of Veeva's Ostro acquisition. Learn how conversational AI integrates with Vault CRM for MLR-compliant HCP and patient engagement.

Review essential features of Anatomic Pathology LIMS, including digital pathology integration, compliance, and workflows for histopathology and cytology.

Examine how Isomorphic Labs uses AlphaFold AI to advance its first computationally designed oncology and immunology therapeutics into clinical trials.

Explore the updated EU GMP Annex 1 guidelines for sterile manufacturing. Learn how to implement a risk-based Contamination Control Strategy (CCS) for compliance

Understand how AI automates regulatory medical writing for CTD Module 2 summaries in IND and NDA submissions, including efficiency gains and compliance risks.

Analyze the AI-enabled precision proteomics market through the Alamar Biosciences $191M IPO, NULISA technology, and the future of protein biomarker detection.

Learn about CDISC SEND datasets for FDA nonclinical submissions. This guide explains SDTM framework alignment, SENDIG rules, and regulatory compliance timelines

Explore Kailera Therapeutics' $625M IPO and GLP-1 obesity pipeline. This analysis covers clinical data, dual agonists, and the 2026 biotech market reopening.

This report examines pharma data lakehouse architecture for R&D. Review how Databricks, Snowflake, and Apache Iceberg manage life sciences and clinical data.

Examine AI clinical study report automation. This guide explains the technical process of converting SDTM and ADaM datasets into Module 5 submission documents.

An in-depth comparison of AI biology foundation models like AlphaFold 3, ESM3, Boltz-2, and Chai-1. Analyze their architectures, benchmarks, and use cases.

Examine the FDA Commissioner's National Priority Voucher (CNPV) pilot program. This guide explains the new 1-2 month accelerated drug review pathway and rules.

Analyze the Phase IIa clinical trial results of INS018_055 (rentosertib), the first AI-designed drug by Insilico Medicine targeting TNIK for IPF treatment.

Analyze the FDA NAM roadmap for animal-free drug approval. Learn how advanced in silico models, AI tools, and organ-on-chip systems replace animal testing.
Track 2026 Big Pharma and hyperscaler AI deals. This report analyzes enterprise cloud partnerships, infrastructure investments, and GxP compliance trends.

Analyze the $1B Merck and Google Cloud partnership. This report explains how agentic AI and Gemini Enterprise apply to pharmaceutical R&D and data workflows.

Analyze the $838M Merck-Infinimmune AI antibody discovery deal. Understand how the Anthrobody screening platform and GLIMPSE language model engineer biologics.

An overview of the $1B NVIDIA-Eli Lilly AI lab, detailing its 5-year compute strategy, DGX SuperPOD infrastructure, and foundation models for drug discovery.

Examine ChatGPT Enterprise admin controls, security settings, and compliance features. This report details SSO, RBAC, data residency, and workspace policies.

Examine how UVA's YuelDesign uses 3D AI diffusion models in drug discovery. Learn how co-designing ligands with flexible protein pockets improves binding.

Analyze the application of generative AI in antibiotic discovery. This report examines the Phare Bio-Basilea partnership and Gram-negative drug development.

Analyze OpenAI's GPT-Rosalind, a biochemical reasoning model for life sciences. Learn how Amgen, Moderna, and Thermo Fisher apply this AI to drug discovery.

Analyze the Chai Discovery and Eli Lilly partnership in AI biologics design. Examine how generative models like Chai-2 impact antibody drug development.

Examine the $50M GSK-Noetik agreement licensing AI foundation models for oncology. Learn how spatial biology and virtual cells advance R&D and drug discovery.

Analyze the Boltz-Pfizer AI drug discovery partnership. This report examines the application of open-source biomolecular foundation models in pharma R&D.

Analyze Gilead's $5 billion acquisition of Tubulis, examining antibody-drug conjugate (ADC) oncology strategy, market trends, and new clinical platforms.

Review China NMPA drug approval pathways, regulatory strategy, and ICH harmonization. Examine priority, breakthrough, and conditional approval processes.

A detailed analysis of the Takeda-Iambic AI drug discovery partnership. Examine the $1.7B deal terms, NeuralPLexer technology, and AI pharma industry trends.

Comprehensive analysis of Roche Navify Clinical Hub, detailing its platform architecture, AI features, oncology tumor board workflows, and trial matching.

Read an analysis of Amazon Bio Discovery, an AWS agentic AI platform integrating biological foundation models and wet-lab workflows for drug development.

Compare Microsoft 365 and Google Workspace for pharmaceutical GxP compliance. Learn how each cloud platform meets FDA 21 CFR Part 11 and data integrity rules.

Analyze Insilico Medicine's Pharma.AI platform for AI drug discovery. Learn how PandaOmics, Chemistry42, and MMAI Gym integrate for target and molecule design.

Review the core requirements for 21 CFR Part 11 compliance in pharma. This guide explains FDA electronic records, signatures, ALCOA+, and system validation.

Guide to pharma AI procurement with comprehensive RFP templates and weighted vendor scorecards. Review FDA compliance, data privacy, and GxP criteria.

Understand FDA Predetermined Change Control Plans (PCCPs) for AI/ML Software as a Medical Device. Review Section 515C, FDA guidance, and implementation.

Examine how fine-tuning foundation models and LLMs improves pharmaceutical R&D and drug discovery. Review Insilico's MMAI Gym methodology and AI benchmarks.

This guide details how bioanalytical LIMS software ensures method validation and regulatory compliance with FDA 21 CFR Part 11, ICH M10, and GLP standards.

An in-depth analysis of the Amazon Bio Discovery platform. Learn how AWS uses agentic AI, biological foundation models, and integrated wet labs for drug R&D.

Examine the Novo Nordisk and OpenAI partnership to understand enterprise AI strategy in pharma. Explore generative AI applications in drug R&D and manufacturing

Analyze the FDA's 2025 AI-driven enforcement of prescription drug advertising. This report details DTC compliance changes, warning letters, and pharma impacts.

Explore how artificial intelligence is applied in pharmaceutical QC labs to automate OOS investigations, real-time batch release, and stability data analysis.

Guide to structuring a board-ready business case for enterprise AI in pharma. Review investment planning, ROI metrics, implementation strategies, and use cases.

Examine the FDA's enforcement of ClinicalTrials.gov reporting mandates, historic compliance gaps among trial sponsors, and AI solutions for data disclosure.

Explore how AI target discovery and graph-based machine learning platforms like bfLEAP address R&D challenges in central nervous system drug development.

Examine clinical evidence generation for AI diagnostic SaMD. This report covers real-world performance studies, FDA guidelines, and post-market surveillance.

Review the $12B wave of Q1 2026 oncology AI deals and pharma M&A. This analysis explains how patent cliffs, CAR-T, and AI drug discovery drive acquisitions.

Guide to designing an AI training program for pharmaceutical teams. Review role-based curriculum development, learning tools, and ROI measurement frameworks.

Analyze how Roche utilizes its NVIDIA AI factory, featuring 3,500 GPUs and Omniverse digital twins, to scale GLP-1 drug manufacturing and optimize pharma R&D.

Examine the 2026 AI digital health landscape. Learn how Big Tech, pharmaceutical companies, and startups compete and collaborate in AI-powered healthcare.

This analysis examines AI in the pharmaceutical supply chain, detailing machine learning applications for demand forecasting, procurement, and logistics.

Review the ICH E6(R3) Good Clinical Practice (GCP) updates, exploring Risk-Based Quality Management (RBQM), new Annex structures, and implementation steps.

Understand ICH Q7 GMP guidelines for active pharmaceutical ingredients (APIs). Review compliance requirements, quality management systems, and FDA regulations.

Analyze the $2.75B partnership between Eli Lilly and Insilico Medicine. Understand how generative AI impacts pharma R&D strategy and drug commercialization.

Examine the role of AI in clinical trial protocol design. This report analyzes how BMS, Faro Health, and Evinova use digital protocols in drug development.

Analyze Earendil Labs' $787M funding and its implications for AI biologics. This educational report examines pharma AI investments and protein drug discovery.

Analyze Anthropic's $400M acquisition of Coefficient Bio. This report details the integration of large language models and AI in pharmaceutical drug discovery.

Learn how the Dotmatics platform integrates AI into life sciences R&D. This report examines Luma architecture, ELN systems, FAIR data, and lab informatics.

Review AI policies and data classification frameworks used in clinical-stage biotech. Learn how to govern trial data and navigate global AI compliance laws.

Understand ChatGPT Enterprise connectors and Office 365, SharePoint, and Azure integrations. This guide explains RAG architecture, security, and governance.

Analyze token usage patterns and optimization techniques for ChatGPT and Claude. Understand LLM context windows, API costs, and prompt engineering strategies.

Analyze the role of AI in pharma and biotech. Learn how machine learning is applied to drug discovery and clinical trials through industry case studies.

Learn how Egnyte AI and the Model Context Protocol (MCP) integrate secure generative AI and RAG into enterprise content management and data governance.

A factual comparison of ChatGPT Enterprise vs Claude Enterprise. Analyze context windows, compliance controls, model capabilities, and enterprise pricing.

Read an in-depth 2026 comparison of leading AI coding assistants: Claude Code, Codex CLI, and Gemini CLI. Review their features, models, and market adoption.

Examine Amgen's phased enterprise AI rollout to 20,000 employees. Learn about the Microsoft Copilot pilot, security guardrails, and AI adoption metrics.

An educational guide detailing prompt strategies for ChatGPT and Claude in biotechnology. Covers prompt engineering techniques, model comparisons, and examples.

Review a comprehensive directory of Claude Cowork training resources. Access official documentation, community tutorials, video courses, and expert workshops.

Learn how OpenAI Codex assists biotech research by automating bioinformatics tasks. Read our analysis of its technical capabilities, use cases, and limitations.

Review regulatory requirements for assembling pharma AI validation evidence packages. Examine FDA and EMA guidelines, credibility frameworks, and GxP rules.

Review how Large Language Models (LLMs) extract adverse drug events from clinical notes. Learn about NLP model accuracy, implementation, and performance metrics

Analyze the FDA CDER 2026 guidance pipeline for artificial intelligence in drug manufacturing and digital health technologies in pharmaceutical development.

An educational analysis of the IQVIA.ai platform, detailing how NVIDIA-powered agentic AI orchestrates pharmaceutical operations and clinical trial workflows.

Explore how pharmaceutical companies use low-code platforms to build GxP-compliant MES and QMS applications while meeting strict 21 CFR Part 11 standards.

Examine the role of agentic AI in pharmaceutical commercial launches. Learn how autonomous multi-agent systems optimize market access and HCP engagement.
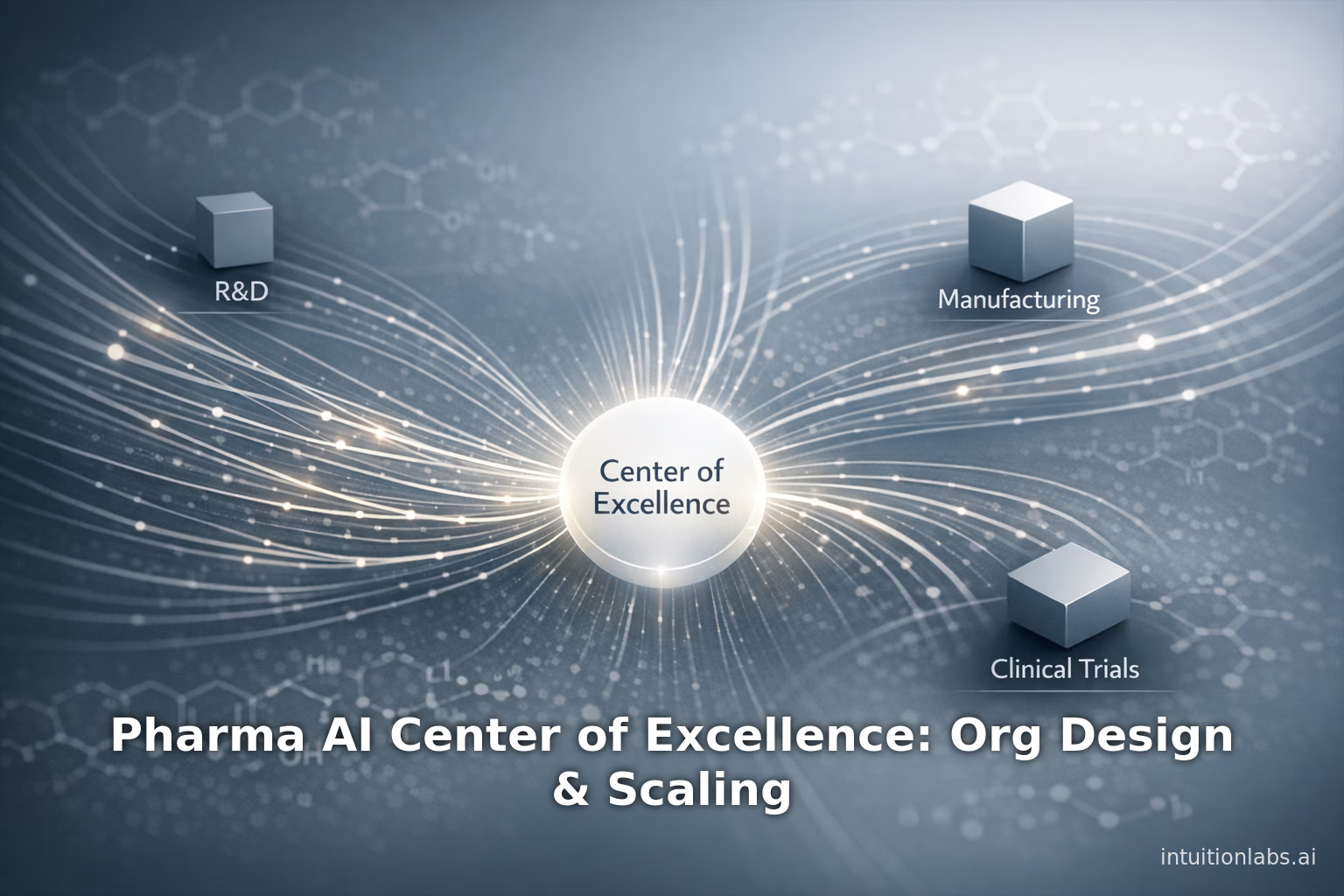
Explore how to structure a Pharma AI Center of Excellence. This guide covers organizational design, hub-and-spoke models, AI governance, and enterprise scaling.

Learn how AI-powered pharma smart factories enable autonomous GMP manufacturing, real-time quality control, and continuous production using digital twins.

Explore SAP S/4HANA implementation in pharmaceutical manufacturing. This guide details GxP compliance, module selection, and computer system validation.

Examine 2026 AI infrastructure investments in pharmaceutical R&D. This report details the Lilly-NVIDIA lab, Earendil funding, and GPU compute deployments.

Examine how pharma organizations deploy private LLMs using air-gapped AI architecture to maintain data sovereignty and comply with FDA and GDPR regulations.

This 2026 guide analyzes GxP compliance software, offering a detailed comparison of eQMS platforms, regulatory standards, and life science quality management.

Examine the 2026 landscape of AI biologics discovery. This analysis details protein modeling, generative design, and key pharma R&D investment trends.

Analyze AI robotics applications in pharma GMP manufacturing. This report details automation use cases across R&D, aseptic fill-finish, packaging, and ROI data.

Read this guide on evaluating digital quality systems (eQMS) for CDMOs. Learn about contract manufacturing software selection, GMP compliance, and integration.

Analyze leading GxP compliance software, eQMS, and document management platforms for life sciences. Learn how systems meet 21 CFR Part 11 and ALCOA+ rules.

Compare ELN, LIMS, and SDMS for pharmaceutical labs. This 2026 guide explains architectural differences, features, compliance, and specific R&D use cases.

Learn how pharma companies build regulatory-grade real-world evidence (RWE) platforms using real-world data for FDA and EMA drug development submissions.

An overview of molecular pathology software, examining LIMS for NGS workflows, multi-omics data integration, and AI analysis tools in precision medicine.

Examine the integration of AI in pharmaceutical CMC submissions and process analytics. Review QbD frameworks, regulatory guidelines, and manufacturing outcomes.

Review NIS2 Directive requirements for the pharma industry. This 2026 cybersecurity compliance checklist covers IT/OT, supply chain, and incident reporting.

Analyze the impact of 2026 U.S. pharma tariffs on global drug supply chains, manufacturing onshoring investments, and digital IT strategy for compliance.

A comprehensive comparison of RTSM software and IRT platforms for clinical trials. Analyze randomization features, trial supply management, and vendor options.

Explore the agentic AI decision framework for the pharmaceutical industry. Learn how to assess build, buy, and partner models for clinical and R&D workflows.

Analyze AI governance in pharmacovigilance signal detection. Review CIOMS XIV and TransCelerate frameworks for risk-based oversight in drug safety monitoring.

Examine how the EU Digital Omnibus impacts pharma AI and medical device compliance. Review AI Act timeline extensions, MDR integration, and GDPR data updates.

Explore a 90-day diagnostic framework for assessing AI readiness in pharma. Learn how to evaluate data governance, regulatory compliance, and integration.

Examine how agentic AI and systems like Microsoft Copilot mitigate the corporate PowerPoint problem in 2026 by automating presentation creation and editing.

Guide to deploying AI in pharmacovigilance under the CIOMS XIV framework. Review core principles for risk-based oversight, model validation, and governance.

Examine the FDA and EMA Good AI Practice guidelines. This comprehensive implementation guide details the 10 regulatory principles for AI in drug development.

Analyze the scientific research capabilities of GPT-5.4 and Claude 4.6. Review how million-token context windows and autonomous tool-use aid data analysis.

Explore Ironclad AI capabilities in contract lifecycle management. This deep dive explains agentic architecture, AI Assist, and generative legal tech tools.

An in-depth comparison of leading pharma MES software: PAS-X, Tulip, MasterControl, and PharmaSuite. Analyze EBR, 21 CFR 11 compliance, and Pharma 4.0.

Explore the architecture and market dynamics of pharmacy management SaaS platforms. Learn about cloud hosting, EHR interoperability, and 2026 growth trends.

Examine how boutique life sciences consulting firms guide pharmaceutical companies through AI adoption, digital health integration, and regulatory compliance.

Understand Computer System Validation (CSV) in pharma. This guide explains GAMP 5 Second Edition implementation, 21 CFR Part 11, and data integrity. Read more.

Analyze enterprise AI admin dashboards and usage controls for ChatGPT and Claude. This guide covers security, compliance, RBAC, and analytics features.

Examine the systemic causes of enterprise AI rollout failures. This report analyzes how poor data readiness, flawed integration, and overhype impact AI ROI.

An analysis of EU AI Act high-risk compliance for pharmaceutical and medical device AI. Covers MDR/IVDR integration, data governance, and 2026 deadlines.

Analyze GLP-1 drug manufacturing dynamics, detailing global capacity expansions, CDMO production strategies, and peptide synthesis tech transfer processes.

Examine HCP Master Data Management in pharma. Review data validation rules, identity verification, and Sunshine Act compliance for aggregate spend reporting.

Review the key changes in IEC 62304 Edition 2 for medical device software. This 2026 guide covers revised safety levels, AI/ML compliance, and expanded scope.

An in-depth comparative analysis of Peer AI and Weave Bio. Examine how these regulatory AI tools utilize LLMs to automate biopharma submission documents.

Examine frameworks for calculating AI ROI in the pharmaceutical sector. Analyze quantitative metrics and payback periods for PV, regulatory, and manufacturing.

Understand the architecture of GxP-compliant data pipelines for pharma AI. This report analyzes data engineering frameworks using Databricks and Snowflake.

Analyze CRO data integration patterns in clinical trials. Learn how sponsors use clinical data lakehouses, APIs, and AI for real-time data independence.

Examine how LLMs like ChatGPT fail in pharma and biotech. Review documented cases of fabricated clinical trials, wrong MOA descriptions, and fake citations.

An analysis of Moderna's enterprise AI adoption strategy. Learn how the company achieved 100% generative AI usage through OpenAI tools and change management.

Analyze the shift from prompt engineering to context engineering in AI. Learn how curating knowledge, memory, and data improves enterprise LLM reliability.

Analyze Dotmatics' evolving natural language query capabilities, AI integration via the Luma platform, and the technical mechanisms of scientific LLM search.

Examine the January 2025 FDA draft guidance on AI in drug development. This report details the 7-step credibility framework and excluded AI applications.

Explore the technical architecture of the Egnyte MCP Server. This guide explains how Model Context Protocol securely connects enterprise data with AI tools.

Examine the enterprise AI knowledge stack. Learn how RAG architecture and tools like Egnyte Copilot turn file repositories into source-grounded AI systems.

Learn how pharma companies transition to AI operating models. Compare Moderna's workforce education, Sanofi's enterprise integration, and BMS's predictive R&D.

Examine how generative AI is transitioning from experimental tools to core AI-native workflow software in legal, regulatory, and clinical research operations.

Examine why enterprise AI demands custom infrastructure, compute, and data pipelines over generic chat apps, featuring the Eli Lilly supercomputer case study.

Explore shadow AI in biotech and life sciences. This report details unsanctioned generative AI usage, data privacy risks, and enterprise governance strategies.

Examine why AI literature review leads biotech R&D with 76% adoption. This report analyzes NLP tools, knowledge extraction, efficiency gains, and future trends.

Compare AI research assistants for drug discovery. Examine how Causaly, Elicit, Consensus, and Semantic Scholar synthesize biomedical literature for R&D.

Examine technical methods for connecting ChatGPT to scientific literature. Learn how RAG pipelines, APIs, and vector databases improve research accuracy.

Learn to design a data layer architecture for AI-powered scientific research. This guide explains FAIR data principles, pipelines, metadata, and storage.

Examine how AI competitive intelligence tools use NLP and machine learning to help biotech BD teams analyze life science data and monitor competitor pipelines.

Compare AI literature mapping tools like ResearchRabbit, Litmaps, and Connected Papers. Learn how visual citation networks aid scientific literature reviews.

Learn how to build a Retrieval-Augmented Generation (RAG) architecture for internal research repositories like ELNs, LIMS, and Egnyte to ground LLM responses.

Examine how 15 biotech startups utilize artificial intelligence to accelerate pharmaceutical R&D, drug discovery, and clinical trials. Read the full analysis.

Learn how to use Elicit AI for structured data extraction from clinical papers. This guide covers LLM workflows, accuracy, and systematic review methodology.

Understand how persistent identifiers connect the research ecosystem. We explain how Crossref, DataCite, ORCID, and OpenAlex link papers, authors, and data.

Examine how AI hallucinations affect drug discovery. Review real examples of LLM errors in pharma R&D and explore practical detection and mitigation methods.

Review free AI tools for PubMed and biomedical literature search. Evaluate AI search engines, evidence-based medicine applications, and current limitations.

Understand the security risks of employees pasting proprietary data into ChatGPT. This guide explores AI data leakage, governance policies, and private LLMs.

Examine the implementation of AI prompt libraries in pharmaceutical workflows. Review prompt engineering techniques, regulatory compliance, and R&D applications

Examine the capabilities of the Miracle clinical operations platform, including automated EDC data integration, AI forecasting, and real-time trial monitoring.

Learn how to deploy ChatGPT Enterprise in regulated GxP environments. This guide covers FDA compliance, 21 CFR Part 11, system validation, and data integrity.

An in-depth analysis of the draft EU GMP Annex 22 guidelines for artificial intelligence in pharmaceutical manufacturing. Review AI compliance and risk rules.

Analyze TransCelerate guidelines for implementing AI in pharmacovigilance. Review a comprehensive roadmap for compliance with FDA and EMA safety regulations.

Examine Bristol Myers Squibb's Predict First strategy for drug discovery. Learn how BMS integrates AI to augment scientific decision-making and R&D efficiency.

An educational analysis of Causaly vs AlphaSense for pharmaceutical competitive intelligence, comparing AI data coverage, R&D applications, and search methods.

Compare citation graph and keyword search methods for finding scientific papers. Learn the limitations, benefits, and how to combine these research techniques.

Examine how AI automates GxP evidence packages and compliance workflows. Review regulatory frameworks, ALCOA+ data integrity, and AI system validation methods.
Explore how Biobank LIMS software enables accurate specimen tracking, complex data architecture, and regulatory compliance for modern biorepositories.

Examine the architectural design and compliance framework for AI governance in pharmacovigilance, including FDA and EU regulatory guidelines for drug safety.

Analyze Moderna's AI Academy to understand enterprise-wide AI upskilling. Learn best practices for corporate AI training, curriculum design, and enablement.

Understand how AI literature review tools function. This guide explains the core architecture, including semantic search, RAG pipelines, and LLM summarization.

Examine how smart manufacturing in pharma uses AI, digital twins, and IoT to optimize production, quality control, and supply chains using a GSK case study.

Explore the transition from traditional keyword queries to AI-driven semantic search in biomedical research. Understand the tools, benefits, and limitations.

Learn to build an AI workflow for research papers using RAG architecture. Move beyond simple PDF chatbots to conduct accurate, systematic literature reviews.

Compare OpenAlex, Semantic Scholar, and PubMed to choose the best academic literature database for your research. Learn their coverage, features, and use cases.

Examine Sanofi's enterprise AI strategy to understand what AI at scale means in pharma. Learn how AI is integrated across biopharma R&D and manufacturing.

Analyze the FDA AI Credibility Framework and its impact on scientific research tools. Review the 7-step risk-based validation process for drug development.

A comprehensive guide to research paper APIs and scientific literature databases in 2026. Learn how to programmatically access scholarly metadata and citations.

Review 10 essential AI tools for pharmaceutical R&D in 2026. This report details platforms for protein structure, generative design, and clinical modeling.

Learn how the Open Scholarly Data Stack uses persistent identifiers like DOIs and ORCIDs to connect research papers, authors, datasets, and citation networks.

Examine responsible enterprise AI practices, including data privacy, misuse prevention, red teaming, and governance frameworks for secure corporate deployment.

This article explains how artificial intelligence, advanced OCR, and LLMs extract unstructured clinical research and CRO data from complex PDF documents.

Explore the root causes of biotech data silos and fragmentation. Learn how to implement FAIR standards and modern data infrastructure in life sciences R&D.

This analysis explains why restricted full-text access and scholarly paywalls are the primary barriers to developing accurate AI tools for research papers.

Examine the pharmaceutical AI strategy roadmap for scaling digital transformation. Learn about enterprise data infrastructure, LLMOps, and GxP compliance.

An in-depth 2026 LIMS software comparison analyzing LabWare, STARLIMS, LabVantage, and Sapio Sciences. Review core features, architectures, and lab use cases.

Review the evolving FDA regulatory framework for AI/ML SaMD. This guide explains PCCPs, total product lifecycle management, and 2026 compliance standards.

Review AI patient recruitment platforms for clinical trials. Compare EHR integration, federated networks, and patient-facing software for trial matching.

Examine top pharma AI consulting firms and life sciences vendors. This 2026 evaluation guide analyzes market trends, FDA regulations, and AI capabilities.

A comprehensive guide to decentralized clinical trials (DCTs). Review hybrid trial technologies, compare software platforms, and explore implementation steps.

Examine how AI and real-time quality monitoring ensure GMP compliance in pharmaceutical manufacturing. Review machine learning frameworks and FDA guidelines.

A guide to FDA regulatory pathways for AI/ML SaMD. Compare 510(k), De Novo, and PMA submission requirements for artificial intelligence medical devices.

Examine how agentic AI automates pharmaceutical regulatory documents like CSRs, NDAs, and eCTD workflows while managing compliance and validation risks.

Learn how to quantify AI ROI in pharmacovigilance. This guide analyzes cost drivers, payback periods, and metrics for automating PV case processing workflows.

Learn why 95% of pharma AI pilots fail to reach production. This guide explains PoC failure causes, data integration challenges, and strategies for scaling.

Explore pharma AI change management and organizational readiness strategies. Understand how life science companies build AI literacy to address skills gaps.

Examine how digital twins in clinical trials function as virtual control arms. This report reviews FDA guidance, AI models, and implementation requirements.

Review the FDA's 7-step AI credibility assessment framework. Understand how to validate and document AI models for regulatory drug development submissions.

Examine the technical and operational factors of CDMS replacement. Compare legacy SAS systems with modern cloud EDC platforms in clinical data management.

Learn how agentic AI in pharma transitions from pilot stages to production. Explore autonomous multi-agent systems, clinical use cases, and regulatory impacts.
Learn how Biobank LIMS software manages specimen tracking, chain-of-custody, and complex data architecture. Compare leading BIMS solutions for biorepositories.

Review how modern CTMS dashboards track clinical trial KPIs. This guide explains operational metrics for enrollment, site performance, and data management.

Examine why most pharma AI pilots fail to reach production. This report analyzes data fragmentation, governance issues, and building scalable data foundations.

A 2026 evaluation framework for selecting pharma AI consulting firms. Review vendor criteria including regulatory compliance, data governance, and AI expertise.

Explore the 2026 FDA-EMA 10 Guiding Principles of Good AI Practice in drug development. Learn about regulatory frameworks, compliance, and AI risk management.

Review a detailed 2026 commercial LIMS comparison. This report analyzes LabWare, STARLIMS, LabVantage, Sapio, and LabWizard features and deployment models.

Review how pharma data engineering workshops train teams in Databricks and AI. Examine Lakehouse architecture, GxP compliance, and workforce skills development.

Analyze IT integration in biopharma M&A. This report explains strategies for consolidating GxP systems, ensuring compliance, and capturing cost synergies.

An analysis of AI governance in pharmacovigilance, detailing how the EU AI Act and GxP regulations impact drug safety platforms like Oracle Argus and Veeva.

Compare open-source MES and ERP solutions for pharmaceutical manufacturing. Analyze ERPNext, Odoo, and others for GMP, 21 CFR Part 11, and Annex 11 compliance.

Review commercial software solutions for GMP-compliant manufacturing. Compare pharma MES, electronic batch records (eBR), and ERP systems for 21 CFR compliance.

Learn frameworks for GxP-safe AI. Covers RAG, audit trails, and data isolation to ensure 21 CFR Part 11 and Annex 11 compliance in pharma.

Review 10 years of FDA 483 patterns and EMA reports. Learn the four core inspection question clusters and the specific evidence required for compliance.

Learn about the impact of AI agents on B2B productivity in 2026. This analysis details enterprise adoption trends, case studies, and Anthropic's technical visio

Explore Project Orbis, the FDA's global oncology review framework. Analyze approval timelines, partner agencies, and challenges in expanding beyond cancer.

Explore Project Orbis, the FDA framework for simultaneous oncology drug review. Understand how international partners coordinate to reduce approval delays.

Examine Project Orbis data on FDA oncology approvals. Analysis shows concurrent review reduced submission gaps to 33 days and accelerated access by 2.6 months.

Analyze how FDA inspection delays and regulatory bottlenecks impact patient safety and drug supply. Learn how AI tools are addressing these critical backlogs.

Learn how FDA QMSR impacts 21 CFR 820. Internal audit and management review records are no longer confidential and subject to inspection under the new rule.

Analyze 2026 strategies for AI-assisted 510(k) submissions. Covers FDA guidance, generative AI tools, and mandatory human oversight protocols.

Analyze the 2026 surge in FDA warning letters for contract manufacturers. Review data integrity risks, regulatory updates, and supply chain oversight strategies

Analyze the FDA QMSR final rule, ISO 13485 alignment, and the new CP 7382.850 inspection playbook. Learn key compliance steps for device manufacturers.

Analyze FDA Project Elsa, a generative AI system that prioritizes facility inspections by detecting risk patterns in adverse events and regulatory data.

Analyze the FDA Quality Management System Regulation (QMSR) effective Feb 2, 2026. Learn how ISO 13485:2016 incorporation impacts medical device compliance.

Analyze the integration of AI with Lean Six Sigma in MedTech. Reviews DMAIC enhancements, regulatory compliance, and predictive quality management systems.

Analyze FDA QMSR requirements for DHF remediation. Learn how to transition legacy Design History Files to the ISO 13485 Medical Device File framework.

Analyze corporate AI training models: Build, Buy, and Hybrid. Review 2026 costs, upskilling strategies, and implementation data for L&D decision-makers.

Guide to drafting a corporate AI policy. Covers governance frameworks, risk mitigation, and compliance with regulations like the EU AI Act and ISO 42001.

Analyze ChatGPT Deep Research features and RAG technology. Understand agentic workflows, automated literature reviews, and accuracy limitations.

Explore the clinical trial regulatory pathway involving FDA, IRBs, and global agencies. Understand approval timelines, compliance steps, and review delays.

Analyze the readiness decay curve and why mock inspections fail. Learn how skill atrophy, CAPA drift, and cognitive biases erode regulatory compliance.

Examine AI hallucinations in business. Learn why LLMs fabricate data, understand the risks, and review technical prevention strategies like RAG.

Learn prompt engineering strategies for business teams. Covers zero-shot, few-shot, and chain-of-thought techniques to optimize AI workflows without coding.

Analyze AI in clinical operations: a guide to workflow automation, scribes, and predictive scheduling. Review use cases, tools, and implementation steps.

Learn to deploy Google Gemini Enterprise. This guide covers agentic AI architecture, data grounding, security protocols, and integration strategies.

Analyze Claude Enterprise architecture, 1M token contexts, and RAG integration. Review 2026 deployment strategies, security protocols, and use cases.

A comprehensive guide to AI data classification policies for ChatGPT, Claude, and Gemini. Learn usage limits, PII restrictions, and enterprise security risks.

Compare 2026 enterprise AI models. Evaluate ChatGPT, Claude, Copilot, and Gemini on security, context windows, and performance benchmarks for business adoption.

Analyze the pharmaceutical AI skills gap and workforce upskilling strategies. Review regulatory impacts, training models, and ROI metrics for R&D teams.

A technical guide to ChatGPT Enterprise deployment. Covers GPT-5 features, data privacy controls, security protocols, and employee training strategies.

Guide to eCTD v4.0 regulatory submissions. Learn about HL7 FHIR standards, document reusability, and global implementation deadlines for FDA and EMA.

Explore the role of interview discipline in FDA inspections. This analysis covers skill decay causes and how AI tools augment auditor questioning techniques.

Compare post-market surveillance for locked vs. continuous learning AI devices. Analyze regulatory guidelines, algorithmic drift, and validation strategies.

Explore AI automation for Clinical Study Reports (CSRs). Analyze efficiency gains, regulatory compliance, and risks like hallucinations and data security.

Analysis of FDA digital health guidance covering SaMD, AI, and cybersecurity. Understand 2026 updates, risk categorization, and regulatory compliance pathways.

Guide to AI-driven literature monitoring in pharmacovigilance. Covers NLP techniques, signal detection, and regulatory intelligence automation strategies.

Explore FDA Structured Product Labeling (SPL) standards, current automation challenges, and how AI integration optimizes pharmaceutical regulatory workflows.

Explore how AI automates CAPA and deviation workflows in Veeva Vault QMS. Covers NLP triage, ML root cause analysis, and regulatory compliance efficiency.

Analyze MLR review automation using Veeva PromoMats AI. Learn how Quick Check and Content Agents reduce compliance errors and streamline approval cycles.

Analyze Veeva Vault CRM for small biotech. Compare features, adoption trends, and learn strategies to right-size your commercial technology stack efficiently.

Analyze Veeva's AI roadmap including the transition from Andi to AI Agents. Covers Vault CRM Bot, AI Shortcuts, and the 2026 implementation timeline.

Learn to validate AI vendor claims in pharma. This due diligence checklist covers GxP compliance, data security, and model performance verification methods.

Explore AI in regulatory writing: analysis of drafting efficiency, clinical study reports, and key risks like hallucinations and data privacy compliance.

Analyze the Veeva-Salesforce split impact on pharma CRM. Learn about required changes to integrations, data migration, and customizations by 2030.

Learn QMS requirements for AI/ML medical devices, including FDA regulations, ISO 13485, data lifecycle management, and model validation strategies.

Explore AI applications in clinical development plans, including protocol optimization, synthetic control arms, and patient recruitment strategies for trials.

A Clinical Development Plan (CDP) outlines the strategy for drug approval. Learn about trial phases, the Target Product Profile, and regulatory requirements.

Learn what PointClickCare EHR is and why it dominates the long-term care (LTPAC) market. This guide covers its core use cases, features, and market share.

Explore the technical and regulatory aspects of Veeva CRM and Epic EHR integration. Covers FHIR standards, HIPAA compliance, and data exchange strategies.

Explore Claude for Healthcare and Life Sciences capabilities. Covers HIPAA-compliant AI, Opus 4.5 integration, CMS data connectors, and drug discovery workflows

An in-depth analysis of the OpenAI Codex app, a command center for AI coding agents. Learn how it enables multi-agent orchestration and parallel workflows.

An in-depth review of inventory software meeting 21 CFR Part 11 FDA rules. Explore key requirements like electronic signatures, audit trails, and ALCOA+ princip

Examine the risks of DIY quality systems in biotech. Learn about FDA Part 11 compliance, data integrity, and when to transition from Excel to eQMS software.

Explore the four layers of a modern biotech software stack—infrastructure, data, apps, and analytics—essential for scaling R&D before Series C funding.

Evaluate TrackWise, MasterControl, Qualio, and QualityOne. Compare biotech eQMS features, FDA compliance, and scalability for startups to enterprises.

Explore the FDA PCCP framework for medical devices. Understand Section 515C requirements for pre-approving AI/ML software changes without new submissions.

Understand FDA SaMD classification for AI/ML devices. Review risk levels (Class I-III), 510(k) pathways, and regulatory guidelines for medical software.

Examine evidence for open-source software in GxP environments. Learn how validation strategies enable compliance while eliminating proprietary vendor lock-in.

Learn 21 CFR Part 11 requirements for electronic records. This guide analyzes cost-effective FDA compliance strategies for small biotechnology startups.

Explore clinical evidence requirements for AI diagnostic tools. Covers FDA/EU regulations, prospective vs. retrospective validation, and performance standards.

Review the ISPE GAMP AI Guide for validating machine learning in GxP. Learn the risk-based framework for data integrity and regulatory compliance.

Explore AI governance frameworks for pharmaceutical companies. Learn to align AI with GxP, FDA regulations, and data integrity standards for safe adoption.

Compare FDA's Computer Software Assurance (CSA) vs CSV for AI systems. Learn risk-based validation strategies for machine learning in life sciences.

Explore GxP requirements for Electronic Batch Records (EBR) in biotech. Covers FDA 21 CFR Part 11 compliance, validation, and current software options.

Analyze when biotechs need pharmacovigilance software. Covers FDA safety database requirements, compliance risks, and solution options for clinical trials.

Explore risk-based AI validation strategies using ICH Q9 guidelines. Learn to manage machine learning lifecycle risks in regulated pharma environments.

Understand EU MDR and AI Act compliance for AI medical devices. Explains classification, conformity assessment, and technical documentation requirements.

Analyze the transition from CSV to FDA's Computer Software Assurance (CSA). This guide details risk-based validation strategies and compliance impacts.

Learn cybersecurity requirements for AI medical devices. Covers FDA guidance, EU regulations, SBOM mandates, and defenses against adversarial AI attacks.

Analyze the life sciences software market projected to reach $45B by 2026. Examine AI trends, key segments, and five structural gaps hindering growth.

Analyze AI integration in pharma IT architecture, from R&D to supply chain. Review key data on MLOps, clinical trial efficiency, and FDA guidance.

Analyze the build vs buy AI decision in pharma. Compare costs, risks, and time-to-value for R&D and commercial teams to guide strategic investment.

A guide to applying ALCOA+ data integrity standards to AI and machine learning. Covers FDA compliance, data governance, and validation for regulated sectors.

Evaluate drug interaction checkers like Lexicomp and Medscape. This review compares sensitivity, clinical accuracy, and features for effective DDI screening.

Explore FDA 21 CFR Part 11 compliance for electronic records and signatures. Learn about validation, audit trails, and data integrity enforcement trends.

Explore GxP audit trail requirements for AI systems. Review 21 CFR Part 11, Annex 11, and ALCOA+ rules for logging training data, prompts, and model outputs.

Explore GxP compliance requirements for ChatGPT and Copilot. Learn about 21 CFR Part 11, system validation, and data integrity strategies for regulated AI.

Compare ICD-10 coding tools from free lookups like icd10data to enterprise CAC systems like 3M. Analyze features, accuracy, and medical coding software.

Explore FDA 21 CFR Part 11 compliance for AI systems. This guide covers validation, audit trails, and data integrity for machine learning in GxP environments.

Learn how AI automates adverse event detection in pharmacovigilance. This guide covers GVP compliance, NLP methods, and validation standards for safety data.

Detailed guide to the Claude Max plan ($100-$200/mo). Analyze 5x and 20x usage limits, feature access, and value for high-volume professional AI workflows.

Explore Mirth Connect architecture, channels, and connectors. Learn how this integration engine handles HL7, FHIR, and DICOM healthcare standards.

Analyze 2026 ChatGPT API pricing, including per-token costs for GPT-5 and GPT-4o. Learn how rate limits and OpenAI usage tiers impact developer budgets.

Analyze global regulatory submissions, contrasting ICH guidelines with regional requirements. Learn to manage FDA, EMA, and local filing differences.

Learn how meta prompting enables LLMs to generate structural scaffolds. Explore recursive techniques, category theory foundations, and efficiency benchmarks.

Learn AI/ML validation in GxP using GAMP 5 2nd Ed. and FDA CSA. Explains risk-based lifecycles, data integrity, and compliance for adaptive models.

Explore the IEC 62304 standard for medical device software. Learn about safety classifications, lifecycle processes, and compliance for FDA and EU MDR.

Explore the Veeva Vault platform for life sciences. Learn about its cloud architecture, GxP compliance features, and applications like Quality, eTMF, and RIM.

A 2026 analysis of AI image generation pricing. Compare Google's Gemini & Imagen costs with OpenAI's GPT Image & DALL-E via API and subscription plans.

Explore the state of AI agents for B2B productivity. This 2026 analysis covers agentic AI, enterprise adoption, economic impact, and Anthropic's solutions.

Learn the key differences in regional eCTD submission guides from the FDA, EMA, & Health Canada. This guide covers Module 1 variations and the shift to eCTD v4.

A guide for emerging biotech on budgeting for Regulatory Information Management (RIM) and eCTD submissions. Covers cost structures, vendor solutions, and compli

An in-depth analysis of eCTD viewer tools for CROs. Explore essential software features, market trends, and preparing for eCTD v4.0 regulatory submissions.

Learn eCTD publishing best practices for compliant regulatory submissions. This guide covers technical standards, validation, and requirements from FDA, EMA, an

Explore eCTD lifecycle management, the core process for updating regulatory dossiers. Learn about ICH standards, submission sequences, and the shift to eCTD v4.

Learn about the ICH M8 eCTD v4.0 submission specification. This guide covers its technical basis in HL7 RPS, file requirements, and global adoption timelines.

Learn the fundamentals of eCTD sequence management for compliant regulatory submissions. This guide explains sequence numbering, lifecycle operations, and best

Explore the complex pricing of eCTD publishing software. Our analysis covers vendor costs, subscription models, and key factors for regulatory submission planni

A comprehensive guide to Records and Information Management (RIM) implementation. Explore typical timelines, key phases, and best practices for regulatory compl

Explore cloud-based regulatory submission tools for pharma. This guide covers eCTD software, RIM systems, compliance (21 CFR Part 11), benefits, and challenges.

Learn how to prepare a Health Canada eCTD submission. This guide covers mandatory requirements, the CESG gateway, validation rules, and the REP process for drug

Compare SaaS vs. on-premise eCTD solutions for pharma. This guide analyzes TCO, scalability, GxP compliance (21 CFR Part 11), and security for both models.

An overview of regulatory submission software for the life sciences. Learn how RIM systems & eCTD publishing tools streamline compliant filings to the FDA & EMA

Learn how to choose eCTD software for small pharma. This guide covers regulatory compliance, global submission timelines for FDA & EMA, and eCTD v4.0 updates.

Learn the most common eCTD validation errors that cause Refuse-to-File (RTF) actions. This guide covers structural, XML, and PDF issues for successful submissio

Learn best practices for managing eCTD submissions. This guide covers the eCTD structure, publishing tools, QC processes, and how to avoid common technical erro

Learn the key differences between the Common Technical Document (CTD) and electronic CTD (eCTD). This guide covers the evolution from paper to the XML-based eCT

Learn the complete eCTD publishing process for regulatory submissions. Updated May 2026 with the latest eCTD v4.0 milestones (PMDA mandate Apr 2026, EMA optional Dec 2025, FDA voluntary, ANVISA 2026).

A complete guide to the IND & NDA eCTD submission process. Learn the eCTD format, CTD modules, FDA requirements, and the transition from eCTD v3.2.2 to v4.0.

Updated May 2026: in-depth comparison of eCTD software for regulatory submissions, analyzing top platforms (LORENZ/MasterControl, Veeva, EXTEDO, Certara) on features, pricing, and eCTD v4.0 readiness.

Learn about eCTD regional variations for global regulatory submissions. This guide compares Module 1 requirements from the FDA, EMA, Health Canada, and more.

Learn the EMA eCTD Gateway specifications for regulatory submissions. This guide explains the technical architecture, AS2 protocols, validation, and file requir

Updated May 2026: Learn about common eCTD validation errors that lead to submission rejection. Covers FDA and EMA rules, eCTD v4.0 rollout, Health Canada Validation Rules v5.3, and strategies for first-pass compliance.

Learn the FDA's mandatory eCTD submission requirements, including key deadlines, technical standards, and the transition from eCTD v3.2.2 to v4.0 for NDAs, BLAs

A guide to regulatory submission quality control (QC). Explore best practices for eCTD dossier checklists to ensure compliance and avoid costly FDA/EMA rejectio

Learn the essential eCTD validation requirements for regulatory submissions to the FDA and EMA. This guide covers technical specifications, business rules, and

Learn the technical requirements for eCTD software submissions to the FDA, EMA, and Health Canada. Updated May 2026 with the latest eCTD v4.0 mandates including Japan's April 2026 cutover.

Learn how integrating eCTD publishing software with a Document Management System (DMS) streamlines pharma regulatory submissions and improves data consistency. Updated May 2026 with FDA eCTD v4.0 voluntary program status and EMA's 2026–2027 v4.0 transition timeline.

A technical guide to eCTD publishing software for regulatory submissions. Compare top tools like LORENZ, EXTEDO, and Veeva for eCTD v4.0 compliance and validati

Explore key strategies for accelerating pharma regulatory submissions. Learn about expedited pathways, process redesign, and the role of AI in shortening drug a

Learn what eCTD viewer software is and why it's critical for regulatory submissions. This guide covers key features, technical capabilities, and tools.

Learn how Regulatory Information Management (RIM) systems help biotech firms manage compliance. This guide details core capabilities, submission planning, and t

Explore key challenges in pharmaceutical regulatory submissions, from legacy eCTD issues and CMC gaps to the rise of AI and structured data for faster drug appr

Learn what Approved WeChat on WeCom is and how it enables compliant pharmaceutical engagement with HCPs in China. Updated May 2026 with Veeva Vault CRM transition (April 2025 GA, Salesforce CRM sunset Sept 2030) and current WeChat MAU figures (~1.39B per Tencent 2024).

Learn about Exeevo CRM for the life sciences industry. This technical guide covers its platform architecture, AI features, and use cases on Microsoft Dynamics 3

An educational analysis of the Syneos Health CRM strategy (updated May 2026 with Syneos's 2023 take-private, Indegene's 2024 IPO, and Veeva Vault CRM migration). Explore its "Dynamic Assembly" model, key tech partners like Veeva & Microsoft, and its impact on HCP engagement.

Learn about m3.com, Japan's dominant physician network with 340,000+ verified MDs (over 90% of doctors). Updated 2026 analysis comparing its CRM and content model to LinkedIn and WebMD.

An educational profile of Close-Up International. Explore its history since 1968, key services in pharma data and CRM, and market position vs. Veeva & Salesforc

Learn to implement deep linking in Veeva MyInsights. This technical guide covers key JavaScript API methods, code patterns for ds.newRecord & ds.viewRecord, and

A technical guide to troubleshooting Veeva MyInsights on iPad. Learn to diagnose and resolve common sync errors, app crashes, and blank dashboards for pharma CR

Complete MyInsights Studio formula syntax guide for Veeva CRM (refreshed May 2026): calculated fields, data elements, query filters, expressions, and X-Pages forward compatibility.

An in-depth guide to the Veeva MyInsights JavaScript API. Learn to use the ds object and core methods like runQuery to query CRM data for custom dashboards.

Updated May 2026. Learn about value-based contracting (VBC) in pharma, including the CMS Cell and Gene Therapy Access Model, IRA negotiated prices, and outcomes-based agreements for Casgevy, Lyfgenia, Beqvez, and Roctavian.

An educational guide comparing the EU Risk Management Plan (RMP) vs. the US REMS for drug safety. Learn about key differences in scope, content, and triggers.

Learn to implement an audit-ready ISO 27001 ISMS for life sciences. This guide covers integrating cybersecurity with GxP, QMS, and FDA regulatory compliance.

Learn the key differences between LIMS, ELN, SDMS, and CDS. This guide explains the purpose, features, and use cases for each lab informatics system.

A clear comparison of the DSUR vs. the PSUR/PBRER. Understand the distinct purpose, scope, timing, and governance of these key pharmacovigilance safety reports.

Learn about patient support hub programs in specialty pharma. This guide covers key workflows, technology, data management, and compliance for improving drug ac

An in-depth guide to HTA dossiers. Explore the evidence requirements, economic models, and submission templates for NICE (UK), CADTH (Canada), and ICER (US).

An in-depth guide to the ICH E2B(R3) standard for pharmacovigilance. Explore the data structure of ICSRs, electronic reporting rules, and key global systems. Updated April 2026.

Compare CareMetx, AssistRx, PHIL, and Lash Group (Cencora) patient hub platforms in 2026 — covering technology, services, performance metrics, and the impact of IRA Part D reforms and the Medicare Drug Price Negotiation rollout.

Analyze leading pharmaceutical artwork management systems. This guide compares Esko, Kallik, and Blue Software for pharma label compliance, workflows, and audit

Choosing a GxP ELN? Compare Benchling, IDBS, and LabArchives on features for 21 CFR Part 11 compliance, system validation, and ALCOA data integrity principles.

Learn the compliant process for answering off-label HCP inquiries. This guide details FDA guidance, medical information SOPs, and software for case management.

Learn how pharma firms use competitive intelligence software to mitigate patent cliff risks. Updated April 2026 with the Humira/CVS Caremark biosimilar shift, Stelara LOE, and AI-native CI platforms.

Efficacy in clinical trials often overstates real-world effectiveness. Learn why this gap exists and how HEOR uses real-world evidence (RWE) to correct for it.

Outcomes-based drug contracts and the IT/data infrastructure needed to track real-world patient outcomes. Updated April 2026 with CMS Multiple Best Prices, CGT Access Model, and IRA impacts.

An educational guide to QALYs and ICERs, the core metrics in health economics. Learn how they measure value and guide cost-effectiveness decisions. Updated April 2026 with NICE's new £25k–£35k/QALY threshold.

An in-depth comparison of ICER (US) vs NICE (UK). Explore their methods, QALY thresholds, and how they assess drug value to influence global pricing floors.

Learn what must be included in a pharma quality agreement. This 2026-updated guide covers key clauses, sponsor vs. CDMO responsibilities, and GMP regulatory requirements including FDA 2016 guidance, EU GMP Annex 1 (2023), ICH Q9(R1), and DSCSA.

Learn the fundamentals of pharmaceutical stability programs. Covers study design, execution, and data trending per the new consolidated ICH Q1 (2024) and FDA guidelines.

Understand analytical method validation with this deep dive into ICH Q2(R2). Explore validation parameters, documentation, and the new QbD lifecycle approach wi

Lifecycle approach to biotech process validation: PPQ and CPV planning, execution, and monitoring under FDA, EMA Annex 1/15, ICH Q9(R1), Q12, and Q13 guidance.

An educational guide to generic drug patent challenges. Learn about the Hatch-Waxman Act, the ANDA pathway, Paragraph IV certifications, the FDA Orange Book, FTC delisting actions, and the 2024 Teva v. Amneal Federal Circuit ruling.

Learn how pharma companies compliantly fund physician education via unrestricted grants. Explore ACCME standards, the Sunshine Act, and firewalls for ethical CM

Learn how Process Analytical Technology (PAT) uses sensors like NIR & Raman for real-time reaction monitoring in pharmaceutical manufacturing to improve quality

Learn how Quality by Design (QbD) and IT systems are shifting pharma from end-product testing to real-time quality assurance with PAT and data analytics.

Updated April 2026: Million-dollar gene therapy pricing analysis covering Casgevy, Lyfgenia, Lenmeldy, Elevidys (with 2025 safety update), the Roctavian and Beqvez market withdrawals, and CMS's 2025 Cell and Gene Therapy Access Model. Compares one-time cure costs to lifetime chronic care using value-based pricing, ICER QALY thresholds, and outcomes-based contracts.

Analysis of how Large Language Models (LLMs) automate clinical evidence collection for HTA economic dossiers. Covers systematic reviews, data extraction, and ch

Learn how patient quality of life (QoL) data is quantified into Quality-Adjusted Life Years (QALYs) to perform cost-effectiveness analysis for specialty drugs.

How real-world data (RWD) and evidence (RWE) validate clinical trial assumptions in post-market economic models. Updated April 2026 with EU JCA, DARWIN EU, FDA finalized RWE guidance, ICH M14 and EHDS developments.

Learn how health economic data like cost-effectiveness analysis is used to secure drug formulary placement and reimbursement from payers and HTA bodies.

Learn the integrated workflow for manufacturing deviations, CAPA, and change control in GMP. This guide covers regulatory requirements and common inspection pit
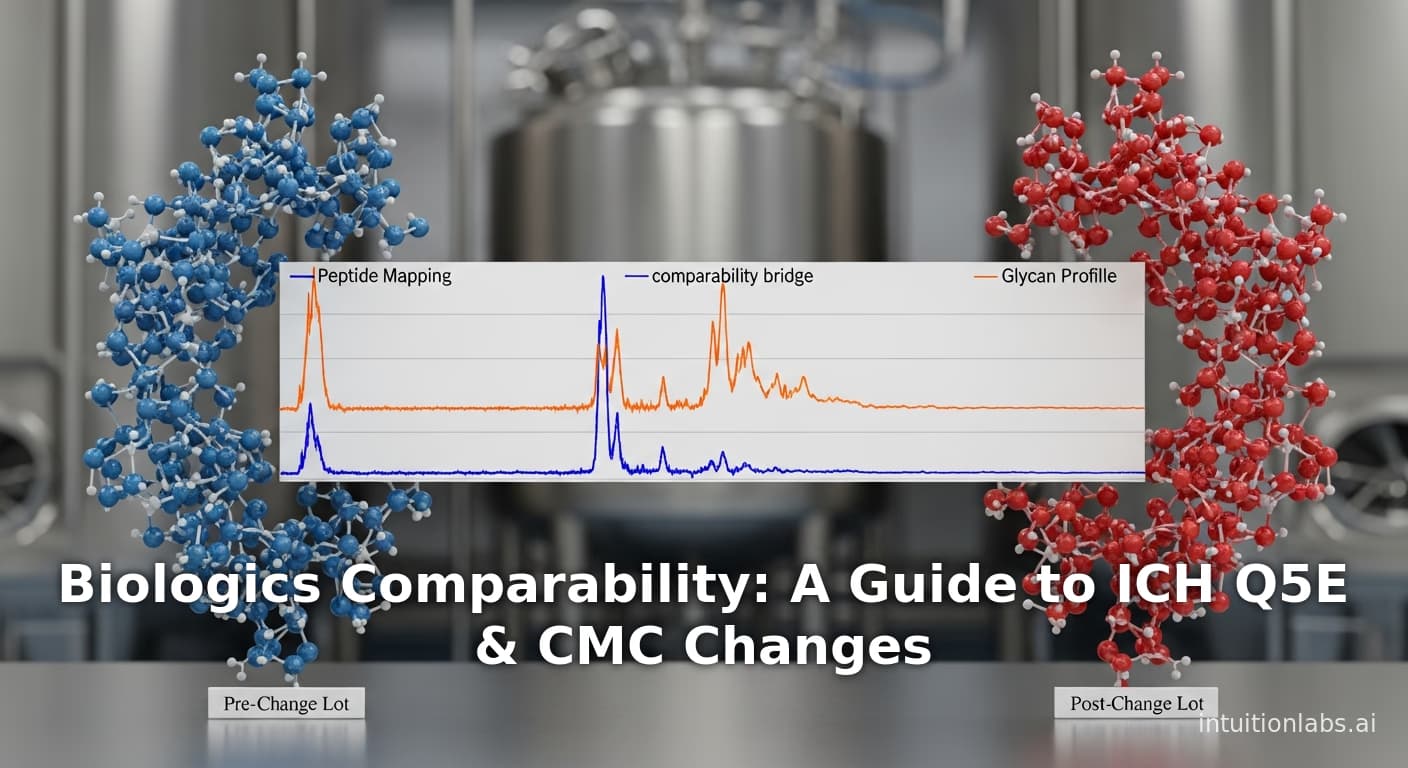
An educational guide to biologics comparability studies. Learn how to prove "no meaningful change" after manufacturing process changes, based on ICH Q5E and FDA

Learn how Integrated Business Planning (IBP) and S&OP help pharma supply chains align demand, capacity, inventory, and constraints. Updated April 2026 with GLP-1 supply lessons, FDA RMP guidance, biosimilar wave 2, and AI in IBP.

Learn how MES and Electronic Batch Records (EBR) improve pharma manufacturing. This guide explains benefits for GMP compliance, data integrity, and review-by-ex

Learn the key differences between US DSCSA and EU FMD for pharmaceutical serialization. This guide covers compliance requirements for ops teams, including data

A proven framework for CDMO selection in pharma and biotech, with scorecards, due diligence checklists, and governance KPIs — updated April 2026 with BIOSECURE Act, Catalent/Novo deal, and EU GMP Annex 1 impacts.

Learn the critical steps for successful pharmaceutical technology transfer. This guide covers the R&D to CDMO process, key documents, and how to avoid scale-up

Learn to validate AI/ML systems in GxP manufacturing using the GAMP 5 Appendix D11 framework. Explore key considerations for data integrity, risk, and model dri

Learn to deploy private LLMs in pharma. This guide covers compliant architecture, including on-premise & cloud, and navigating HIPAA, GxP, and FDA regulations.

Learn how Generative AI (GenAI) applies to Medical Affairs in pharma. This guide covers key use cases, compliance guardrails, and the risks of using LLMs.

Learn about the FDA's AI guidance for drug development. This article explains the 7-step credibility framework, context of use (COU), and risk-based approach.

An in-depth guide to Veeva X-Pages development. Learn how to create custom, contextual dashboards in Vault CRM using HTML, JavaScript, and the DataService API.

Explore Andon Labs' profile and its Project Vend collaboration with Anthropic. Learn how autonomous AI agents using LLMs are benchmarked for business tasks.

Get a detailed list of biotech companies in Toronto (2026). Learn about the life sciences ecosystem, top startups in AI and genomics, and key industry players.

An in-depth analysis of the build vs. buy decision for a quality management system (QMS) in biotech startups. Explore costs, timelines, and compliance factors.

Navigate FDA 21 CFR Part 11 for AI systems. This article details compliance strategies for validation, audit trails, and data integrity in regulated GxP setting

Learn to apply the GAMP 5 risk-based approach for AI/ML validation in GxP environments. This guide covers data integrity, model drift, and regulatory compliance

Explore how AI Contract Lifecycle Management (CLM) software streamlines biotech contract workflows, ensures regulatory compliance, and accelerates clinical tria

Compare the IEC 62304 standard vs. FDA CSA guidance for medical software. Learn key differences in scope, risk classification, validation, and documentation.

Understand the FDA's final Computer Software Assurance (CSA) guidance, a risk-based shift from CSV for pharma & biotech. Learn key principles for compliance.

Learn how to integrate GAMP 5 (2nd Ed.) and FDA's final Computer Software Assurance (CSA) guidance (Sept 2025, updated Feb 2026). Shift from CSV to risk-based validation.

An evidence-based analysis of 20 biotech startups poised for impact by 2026. Learn about key innovations in gene therapy, diagnostics, AI, and sustainability.

A comprehensive analysis of all n8n alternatives. Compare SaaS (Zapier, Make), open-source, and AI-native platforms for your next workflow automation project.

Nvidia paid $20B for Groq's assets (2.9x its $6.9B valuation). Analysis of the deal structure, LPU technology, and antitrust implications — updated April 2026 with the Warren-Blumenthal Senate inquiry.

An in-depth analysis of Montreal's biotech ecosystem (updated April 2026). Key companies, startups, research institutions, and the economic drivers of this top life sciences hub — including the latest funding rounds, clinical milestones and the Quebec Life Sciences Strategy 2025–2028.

Learn about the Turing machine, the foundational model of computation in computer science. This guide explains its definition, components, and the Church-Turing

Learn how to build SAP dashboards for pharma and biotech. Covers SAP Business Data Cloud, Analytics Cloud with Joule AI, Datasphere, KPIs, and GxP compliance (2026).

An analysis of Alphabet's $4.75B acquisition of Intersect Power, which closed March 10, 2026. Learn why Google bought the energy firm to solve AI's massive data center power demand.

Learn about the NVIDIA GB200 supply chain. We analyze the massive global ecosystem of hundreds of suppliers required, from TSMC's silicon to HBM3e and CoWoS pac

Learn the key differences between High Bandwidth Memory (HBM) vs. DDR. Explore HBM's 3D architecture, HBM3E/HBM4 generations, bandwidth benefits over DDR5, and its role in AI & HPC GPUs (NVIDIA Blackwell, AMD MI350).

Learn about DDR6, the next-gen memory standard. We explain its 17,600 MT/s speeds, new 4x24-bit channel architecture, and how it compares to DDR5 for AI & HPC.

An in-depth analysis of Coursera's $2.5B acquisition of Udemy, now approved by shareholders (April 2026). Learn about the merger's impact on the online learning market, AI reskilling, and the future of EdTech.

An educational summary of late 2025 AI research papers. Learn about advances in foundation models like GPT-5, agentic AI, and new neuromorphic hardware.

Learn what reinforcement learning (RL) is through clear explanations and examples. This guide covers core concepts like MDPs, agents, rewards, and key algorithm

Learn what SharePoint Framework (SPFx) is, the client-side model for M365 development. Explore its history, architecture, and use with AI coding agents like Cla

A technical analysis of Google TPU architecture, from v1 to v7. Learn how this custom AI accelerator powers Gemini 3 with superior performance and efficiency vs

Learn why MSLs can discuss off-label data via scientific exchange while sales reps cannot, including FDA's 2023 SIUU guidance on firm-initiated scientific communications.

An analysis of private equity value creation for TPA & PBM companies post-acquisition. Learn key strategies for growth, operational efficiency, and M&A integrat
2026 guide to Veeva Vault CRM X-Pages development services. Covers X-Pages Studio vs custom code, Veeva AI Agent integration, Nitro data, timelines, costs, and how to select a certified development partner.

Learn the 5-stage eQMS maturity model for Biotech & MedTech startups. Updated April 2026 for the FDA QMSR (21 CFR 820 + ISO 13485:2016) effective Feb 2, 2026, CDER's third-year QMM pilot, and the latest Quality 4.0 AI trends.

This 2026 analysis reviews top patient payment platforms for automating collections. Compare features and performance of PatientPay, Cedar, RevSpring, and more.

Explore a detailed comparison of cash flow forecasting software for health systems. This 2026 analysis reviews StrataJazz, Obol, and Centage for hospital financ

A data-driven analysis of 2025 KLAS scores for top enterprise PACS vendors. Compare Sectra, Agfa, Fujifilm, and others on user satisfaction and performance.

Compare top healthcare CMMS platforms. This analysis reviews Accruent TMS, Brightly Biomed, and TRIMEDX RSQ on features, compliance, ROI, and user experience.

Learn the SCDC framework for differentiated messaging of high-value AI solutions in life sciences. This guide explains how to tailor content for diverse stakeho

Explore a 2026 ROI analysis of Quality 4.0 in pharma manufacturing. Learn how technologies like AI & digital twins drive up to 45% cost savings and 40% capacity

Learn what a Case Report Form (CRF) library is and how it improves clinical trials. Covers CDASHIG v2.1, ODM-XML v2.0, Veeva Vault EDC, Medidata Designer AI, and the FDA Dataset-JSON RFC (Apr 2025).

Explore OpenStudyBuilder, the open-source platform for clinical study specifications. Updated for 2026 with ICH M11 adoption, CDISC 360i launch, USDM 4.0, and growing multi-company collaboration.

Updated April 2026 for ServiceNow's Yokohama and Zurich releases, the Moveworks acquisition, and new FDA/EMA AI guidance. A guide to ServiceNow use cases in healthcare and life sciences – ITSM for clinical workflows, patient experience, HIPAA/GxP compliance, EMR integration, and agentic AI.

An in-depth analysis of OpenAI's GPT-5.2 breakthrough on ARC-AGI-2 (54% in Dec 2025), the rapid progress to 98% by April 2026, and the launch of ARC-AGI-3 that humbled all frontier models back to near-zero.

Explore the modern biotech lab's evolution in 2026. Learn how automation, AI, self-driving labs, and digital tools are transforming research, from high-throughput screening to autonomous discovery platforms.

An analysis of the Agentic AI Foundation (AAIF) by the Linux Foundation. Updated April 2026 with 170+ members, MCP's 110M+ monthly downloads, A2A v1.0, and enterprise adoption stats.

Explore document automation for pharma in 2026. Learn how template-driven assembly, structured content, and agentic AI can cut CSR authoring time by 40-90%, with real-world case studies from Merck, Narrativa, and more

Learn how Retrieval-Augmented Generation (RAG) connects ELN, LIMS, and institutional knowledge for drug discovery. Updated for 2026 with FDA/EMA AI guidelines, Benchling AI agents, GraphRAG, and latest market data.

Learn how machine learning (ML) and AI are transforming pharmaceutical CMC process optimization in 2026. Covers applications, digital twins, regulatory updates (FDA/EMA 2026 AI principles, FRAME initiative), and real-world case studies.

Learn how generative AI and LLMs assist in drafting pharma IND & CTA submissions. This guide covers benefits, risks, GxP compliance, FDA/EMA 2026 joint AI principles, and tools like AutoIND and Narrativa.

Learn to build an AI-powered competitive intelligence stack for biotech in 2026. Covers data sources, NLP/ML methods, generative AI, agentic workflows, EU AI Act compliance, and use cases for R&D strategy.

An educational guide to making your biotech AI-ready. Explore essential data infrastructure fixes for data quality, integration, compute, and governance — updated for 2026 with the latest on pharma supercomputers, FDA/EMA AI guidance, and industry best practices.

Learn how to measure AI ROI in drug discovery beyond cost and time savings. Updated for 2026 with 173+ AI clinical programs, Insilico's Phase IIa results, Recursion-Exscientia merger, and new FDA guidance. Key metrics for scientific outcomes, pipeline productivity, and commercial value.

A framework for biotech CDOs on the build vs. buy AI decision. Analyzes costs, time-to-value, integration challenges, vendor evaluation criteria, and the emerging foundation model licensing trend. Updated April 2026 with EU AI Act compliance deadlines, FDA-EMA joint guidance, and latest pharma AI partnerships.

Updated 2026 guide to AI-assisted SOP drafting in GxP life sciences. Covers FDA CSA final guidance, FDA-EMA GxP-AI Principles, EU AI Act compliance, EMA Annex 22, and validated AI platforms for pharmaceutical quality management.

Explore the ROI of predictive maintenance for lab instruments. Updated for 2026, this guide analyzes ML scheduling cost-benefit factors to reduce downtime and extend equipment life

Learn how AI, LLMs, and agentic AI can build a "Second Brain" for pharma R&D. Updated for 2026 with GraphRAG benchmarks, FDA AI guidance, and enterprise deployment patterns for searchable institutional memory in drug discovery.
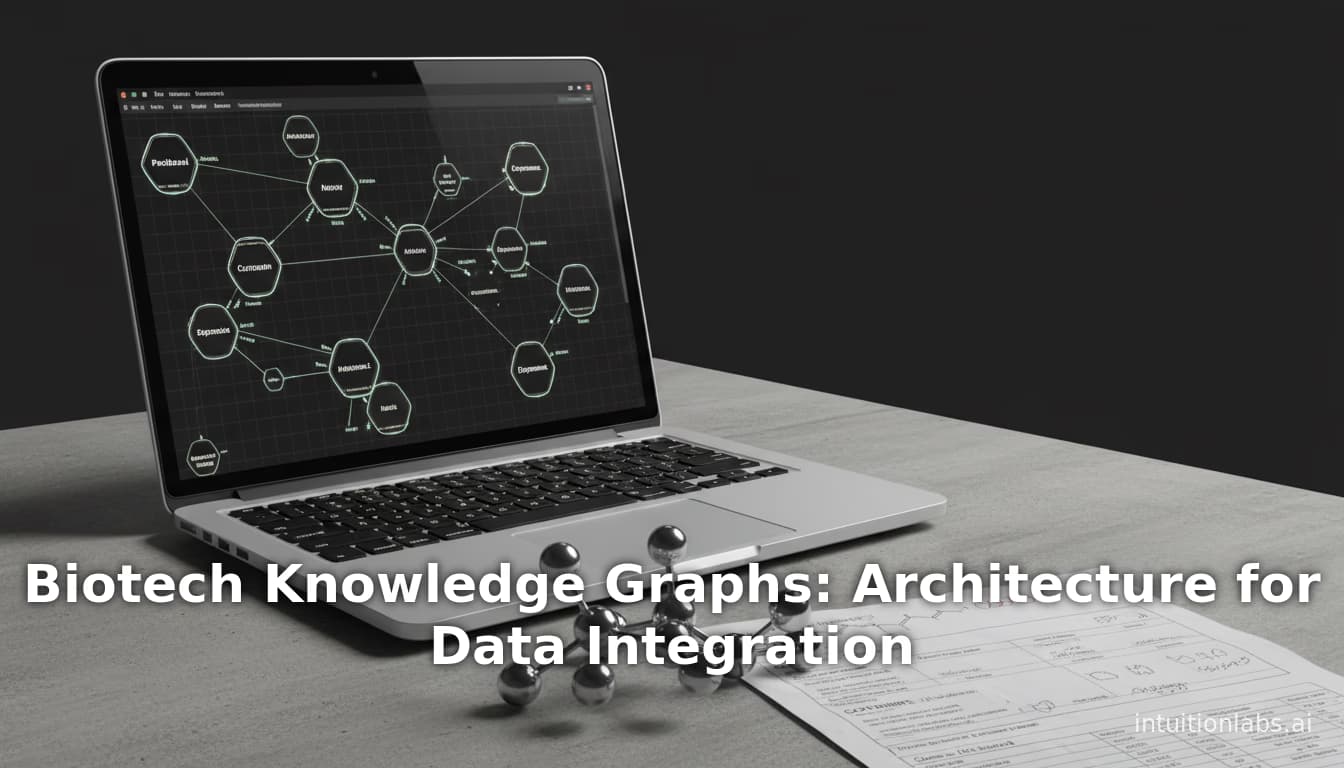
Learn how biotech knowledge graphs are built for drug discovery. This guide covers architecture patterns (ETL, ELT, GraphRAG) for integrating compounds, assays, and targets, with 2026 updates on LLM integration and FAIR standards.

Learn how Large Language Models (LLMs) like GPT-5.4 and RAG pipelines are used for financial document analysis. Updated for 2026 with benchmarks, SEC AI disclosure rules, and agentic workflows for extracting insights from SEC filings and corporate decks.

An in-depth guide for bench scientists on using LLM copilots in research. Covers GPT-5.2, Gemini 2.5 Pro, Benchling AI, DeepMind co-scientist, real-world performance data, FDA guidance, and 2026 adoption trends.

Understand algorithmic redlining, a form of AI bias creating housing and lending discrimination. Explore its causes, legal frameworks including the EU AI Act and state enforcement, and effective technical solutions. Updated with 2025-2026 enforcement actions, CFPB changes, and Colorado AI Act developments.

Learn why spreadsheets fail for life sciences contract management. This guide explores how AI-powered CLM platforms — including agentic AI tools like Icertis Vera — improve efficiency, compliance, and clinical trial cycle times. Updated for 2026 with EU AI Act enforcement, FDA/EMA joint AI principles, and the latest Gartner Magic Quadrant CLM leaders.

Compare per-token API costs: Grok from $0.20/M, Gemini $1.25/M, GPT-4o $5/M, Claude Opus $15/M. Updated pricing tables and enterprise plans.

An educational overview of the NeurIPS 2025 conference. Learn about key trends in AI research, including LLMs, major awards, acceptance rates, and new paper tra

An analysis of the 2025 RAM shortage. Learn how massive AI demand for HBM is straining the DRAM supply, leading to surging prices for PC, gaming, and server mem

NVIDIA H100 costs $25K-$40K, B200 $30K-$50K, DGX B300 $300-350K. Compare H100, H200, B200, B300 purchase vs. cloud rental costs with full 2026 pricing breakdown.

A complete guide to Anthropic Claude pricing in 2026. Learn about subscription plans (Pro, Max, Team) and per-token API costs for Opus 4.6, Sonnet 4.6, and Haiku 4.5, plus Managed Agents and tool pricing.

Updated April 2026 guide to Microsoft Copilot pricing ($30/user add-on, $99 E7 Frontier Suite), licensing changes, April 2026 paywall updates, and qualifying Microsoft 365 plans.

A complete guide to Google Gemini for business plans and pricing (updated April 2026). Learn about Gemini 3.1 Pro, AI Pro/Ultra subscriptions, API costs, Workspace tiers, enterprise models, and real-world use cases.

An in-depth guide to Mistral Large 3, the open-source MoE LLM. Learn about its architecture, 675B parameters, 256k context window, benchmark performance, and 2026 ecosystem developments including Forge, Small 4, and Voxtral TTS.

Learn about Causaly Pipeline Graph and Agentic Research, AI platforms integrating competitive intelligence and knowledge graphs for pharma R&D. Updated April 2026 with market data, FDA guidance, and platform evolution.

Learn about Clarivate Cortellis, the life sciences intelligence platform for drug discovery and R&D. Updated for 2026 with AI assistant launch, Claude MCP integration, and potential divestiture news.

An in-depth 2026 review of the AlphaSense market intelligence platform. Learn how its AI search, Generative Grid, Financial Data, and Workflow Agents work, plus user reviews, $500M ARR growth, and competitive analysis.

An in-depth analysis of the Los Angeles biotech industry (updated April 2026). Covers ~4,000 life science companies, $60B+ economic output, Amgen's $600M expansion, Capsida's IND clearances, UCLA Research Park, and NIH funding developments shaping LA's ecosystem

An in-depth review of top AlphaSense alternatives for market research in 2026. Compare features & use cases of Bloomberg, FactSet, PitchBook, Klue, and expert networks

Updated 2026 guide to the Veeva Direct Data API. Covers features, open-source accelerators for Snowflake/Databricks/Redshift, 26R1 Vault Time changes, AI Agents integration, and bulk data extraction for analytics and AI on Veeva Vault data.

Explore the Boston-Cambridge biotech ecosystem in 2026. This guide covers top companies like Biogen, Vertex & Moderna, venture funding trends, federal policy impacts, and Massachusetts' drug development pipeline.

Explore San Diego's biotech hub, a top 3 U.S. cluster. Updated for 2026 with $4.8B VC funding data, NIH funding crisis impact, major acquisitions (Avidity $12B, Poseida $1.5B), and company updates for Illumina, Neurocrine, Halozyme

Learn how AI and machine learning analyze Real-World Data (RWD) to generate Real-World Evidence (RWE). Explore key applications including LLM-driven data extraction, pharmacovigilance, and synthetic control arms, plus 2025-2026 FDA and EMA regulatory updates.

An in-depth analysis of the $300B Oracle-OpenAI cloud computing deal, updated through April 2026. Covers the operational Abilene flagship, $122B OpenAI funding round, Oracle's $523B backlog, Stargate expansion to 7 GW, and IPO preparations.

Explore a data-driven analysis of AI's impact on the 2025 job market for recent graduates. Learn about declining entry-level jobs and new skill demands.

A technical review of Gemini Nano Banana Pro and Nano Banana 2, Google’s AI image models. Learn key specs, pricing, 1M-token context, and applications in life sciences

Explore practical applications of Claude Code in life sciences. This guide covers its use in genomics, bioinformatics, and R&D automation via tool connectors. L

A data-driven analysis of the 2025 Clinical Research Associate (CRA) job market, covering global demand, salary benchmarks, turnover rates, and career outlook.

Learn best practices for eDiary data collection in clinical trials. Updated for 2026 with ICH E6(R3), FDA DCT guidance, AI integration, and eCOA vendor landscape changes.

Explore 2025 data on AI adoption in private medical practices. Learn how AI tools for administrative tasks reduce burnout and see key stats on usage and gaps.

Analysis of OpenAI's ChatGPT ads strategy — now live since February 2026. Covers the financial data, 900M+ weekly users, $25B revenue, Go tier, IPO plans & AI monetization

Analyze the 2025 AI engineer job market. Learn the core responsibilities, average salary, and how the role differs from a data scientist or ML engineer.

A technical overview of Claude Opus 4.5 and 4.6, state-of-the-art AI models for coding. Learn their capabilities for software development in healthcare and pharma. Updated February 2026.

Comprehensive guide to the EU Falsified Medicines Directive (FMD) and global pharma serialization. Covers DSCSA 2025 enforcement, EMVS verification, GS1 barcodes, track-and-trace compliance, and 78+ country mandates.

Explore how GenAI helps manage pharmaceutical labeling in 2026. Learn to draft local label deviations from a CCDS using AI tools like Veeva AI Agents, and understand FDA-EMA guidance on AI in regulatory compliance.

Who owns pharmaceutical proofreading? Updated for 2026 with EU ePI mandates, DSCSA enforcement, Veralto-GlobalVision acquisition, and AI-augmented labeling workflows across artwork, regulatory, and QA teams.

Learn the end-to-end pharmacovigilance process for translating safety signals into drug label updates. Covers CCDS, Vault Signal, EU Regulation 2025/1466, FDA AEMS, and how modern PV platforms accelerate the safety-to-label relay. Updated April 2026.

Explore the career path from a Regulatory Affairs Generalist to a specialized Labeling Strategist. Learn key skills, role differences, salary data, and 2026 industry trends including mandatory EU ePI and digital labeling.

A comprehensive guide to the Target Product Profile (TPP) for drug development, updated for 2026. Learn TPP components, FDA and ICH guidance, IQVIA adoption data, and how to align your TPP with the final drug label.

Learn how Veeva RIM automates end-to-end pharmaceutical labeling. This guide explains 5 core workflows for label change management and regulatory compliance, updated with 2025–2026 platform enhancements, AI Agents roadmap, and EU ePI developments.

Get a 6-month checklist for pharmaceutical labeling operations. Updated for 2026 with DSCSA enforcement deadlines, FDA PMI rule status, EU ePI developments, and current recall data.

Learn how Veeva PromoMats' Auto-Update Anchors and new AI Agents automate marketing claims substantiation to ensure regulatory compliance and reduce manual MLR review effort. Updated for 2026 with latest Veeva AI capabilities and FDA/FTC enforcement trends.

A guide to Reference Safety Information (RSI) in clinical trials. Learn the regulatory framework including ICH E6(R3), EU CTIS, and UK 2026 CT Regulations, common pitfalls, and how to avoid SUSAR reporting errors.

Updated 2026 guide to Structured Product Labeling (SPL) and data integrity. Covers FDA mandates, EMA ePI roadmap, Health Canada XML-PM, FHIR standards, and ALCOA+ compliance for pharmaceutical labeling.

Explore the regulatory process for negotiating an FDA boxed warning on drug labeling, from initial NDA review to post-market Safety Labeling Changes (SLCs), with 2025-2026 case studies including HRT warning removal, Elevidys, Carvykti, and COVID vaccine labeling debates.

Learn to write the Highlights of Prescribing Information (HPI). Updated for 2026 with AI labeling tools, the Prescription Information Modernization Act, and generic drug labeling reforms. Covers FDA requirements, PLR, and best practices.

Learn how the FDA's PLLR replaced old pregnancy categories. Updated 2026 guide on drafting clear pregnancy risk summaries for biologics, with ICH E21, IMPACT trial, and PIANO registry data.

This comprehensive 2025 guide details e-labeling (e-IFU) regulations worldwide. See our country-by-country analysis of QR code acceptance for medical devices &

An evidence-based review of NLP in regulatory labeling (updated April 2026). Learn how AI assists medical writers in drafting Prescribing Information (PI/SmPC), including FDA Elsa, FDA-EMA joint AI principles, and current limitations.

A comprehensive guide to transitioning from XML-based SPL to HL7 FHIR for e-labeling. Updated for 2026 with EMA go-live timeline, Jordan mandate enforcement, FDA PQ/CMC STU2, and latest FHIR adoption data.

Updated 2026 guide to FDA off-label promotion regulations for MSL teams, covering the January 2025 SIUU guidance, September 2025 OPDP enforcement surge, USPI boundaries, and compliance best practices

Learn how pharmaceutical translation errors in SmPCs and patient leaflets lead to drug recalls, regulatory sanctions, and patient safety risks. Updated with 2025-2026 EU pharma legislation reform, EMA ePI roadmap, and AI translation developments.

Explore a structured cascade model for rolling out pharma safety labeling updates to global affiliates, covering FDA 505(o)(4) guidance, EMA variations framework, AI-powered PV tools, and sub-90-day implementation strategies.

Updated April 2026 analysis of Google's Gemini 3/3.1 Pro AI for healthcare, pharma, and biotech. Covers MedGemma 1.5, HIMSS 2026 deployments, Isomorphic Labs trials, and FDA/EU regulatory developments.

Learn the difference between a Company Core Data Sheet (CCDS) and local labels. Explore strategies for managing global labeling deviations, including EMA ePI, IDMP compliance, and AI-powered labeling tools for regulatory compliance

Learn the definitions and key differences between basket, umbrella, and platform trials. This guide explains how master protocols work in precision oncology, including 2025-2026 updates on tissue-agnostic approvals, NCI successor trials (ComboMATCH, MyeloMATCH), and evolving FDA guidance.

Updated for 2026: Compare ISO 14155:2026 for medical device trials vs ICH E6(R3) GCP for pharma. Covers scope, risk management, regulatory compliance, and the l

An in-depth guide to adaptive trial design, updated for 2026. Covers ICH E20 guideline, FDA Bayesian guidance, AI-powered trial optimization, and how prespecified changes based on interim data make clinical trials more efficient and ethical.

Learn what Investigator-Initiated Trials (IITs) are and the regulatory responsibilities clinicians face as a Sponsor-Investigator in academic clinical trials, including ICH E6(R3) GCP updates, EU CTIS requirements, and 2025-2026 regulatory changes.

An educational guide to sample size calculation in clinical trials. Learn the roles of statistical power, effect size, and alpha/beta errors in trial design.

Updated 2026 analysis of cell and gene therapy logistics for clinical trials, covering cryogenic cold chain, chain of identity, decentralized manufacturing, and the latest regulatory developments from FDA, EMA, and ICH

Explore the Bid Defense Meeting (BDM), a critical step for CROs to win clinical trial contracts. Updated for 2026 with ICH E6(R3) guidance, AI-driven bid tools, and current CRO market data. Learn key preparation strategies, sponsor criteria, and best practices.

Explore the rise of CRO consolidation in clinical trials, updated for 2026. Covers major M&A deals (Thermo Fisher/Clario, Fortrea spin-off, Syneos take-private), market trends, AI-driven acquisitions, and the impact on pharma sponsors and trial quality

This guide defines Fair Market Value (FMV) for clinical trial investigator compensation. Learn key regulations like the Anti-Kickback Statute and Stark Law to e

Updated 2026 guide to entry-level clinical trial roles. Covers Clinical Trial Assistant (CTA) and IHCRA positions, responsibilities, salary ranges ($47K-$72K), ICH E6(R3) impact, and career paths.

Learn how MedDRA (v29.0) and WHODrug (688K+ products) standardize coding for adverse events and medications in clinical trials. Covers structure, AI-assisted coding, regulatory requirements, and 2026 updates

Analyze the future of Contract Research Organizations (CROs) to 2030. Learn about market growth forecasts, the impact of AI, and decentralized clinical trial mo

Explore the evolution from paper CRFs to eCRFs for clinical trials. Compare paper vs. electronic data capture (EDC) on data quality, cost, time, and compliance. Updated for 2026 with ICH E6(R3), AI-powered eCRF design, decentralized trials, and the latest EDC market trends.

Learn about the database lock process in clinical trials. Updated for ICH E6(R3) 2025. Covers data integrity, soft vs hard lock, AI-accelerated cleaning, RBQM compliance, and best practices

Learn the key factors CROs use for clinical trial site selection. This guide covers patient recruitment, feasibility assessments, and data-driven methods for su

Learn the complete query management process in clinical trials. This guide covers the workflow, high costs, and impact on data integrity for CROs and sites.

Explore the end-to-end Request for Proposal (RFP) process for clinical trial outsourcing. Learn best practices for sponsors to write and evaluate CRO bids, updated with ICH E6(R3) guidelines and 2025-2026 market data.

A detailed analysis of clinical trial start-up costs, from IRB fees to site activation. Explore 2025-2026 benchmarks, hidden expenses, AI-driven cost reduction, and budgeting strategies for sponsors and CROs

Compare ACRP vs. SOCRA clinical research certifications (updated April 2026). Covers ICH E6(R3) transition, eligibility, exam content, updated fees, and recertification for CCRC, CCRA & CCRP

Learn how to manage protocol deviations in clinical trials. Updated for ICH E6(R3) and FDA 2024 draft guidance, covering classification, reporting, and prevention for data integrity and GCP compliance.

Learn the key differences between an Adverse Event (AE), Serious AE (SAE), and SUSAR. Updated for 2026 with ICH E6(R3), EU CTR 536/2014 CTIS, FDA Dec 2025 guidances, and ICH E2B(R3) mandate. Covers definitions, causality, and expedited reporting timelines.

Learn what an Investigator's Brochure (IB) is, its required content per ICH GCP E6(R3) guidelines (finalized 2025), and its critical role in assessing risk for clinical trials.

Learn the essential CDISC standards for clinical trial data. This guide explains SDTM and ADaM data models, their structure, regulatory requirements, and 2025-2026 updates including SDTM v3.0, Dataset-JSON, and ICH M11.

Learn what decentralized clinical trials (DCTs) are, how they work, and the technology involved. Updated for 2026 with ICH E6(R3), FDA/EMA guidance updates, Trials@Home RADIAL results, and current market data. Covers benefits, challenges, regulatory landscape, and future of DCTs.

An evidence-based comparison of digital vs. traditional clinical trial recruitment updated for 2026, covering AI-powered patient matching, DCT market growth, FDA guidance, and hybrid recruitment strategies for diversity.

Prepare for FDA BIMO inspections with this updated 2026 guide for clinical sites & CROs. Covers ICH E6(R3), finalized BIMO guidance, RRAs, documentation, QMS, staff training, & GCP compliance.

Learn about the EU Clinical Trial Regulation (CTR) 536/2014 and the CTIS portal. This guide covers harmonized submission processes, key timelines, and transpare

Explore the critical role of an IRB/IEC in protecting human subjects. Learn how ethics committees review and approve clinical trials per key ethical regulations including ICH E6(R3), the 2024 Declaration of Helsinki, and EU CTIS requirements

Learn why patient retention is critical for clinical trial validity. Updated for 2026 with FDA DCT guidance, ICH E6(R3), AI engagement platforms, and evidence-based strategies to reduce dropouts

A detailed guide to the 13 principles of Good Clinical Practice (GCP). Understand the ICH E6(R2) and finalized E6(R3) standards for protecting subjects and ensuring data integrity.

Learn about clinical trial vendor management and how CROs ensure oversight of labs, logistics, and translation services per ICH E6(R3) GCP and regulatory standards. Updated for 2026 with the latest on ICH E6(R3) implementation, decentralized trial logistics, and AI-assisted translation.

Learn about informed consent forms (ICFs) in clinical trials. Updated for ICH E6(R3) 2025, eConsent innovations, and FDA/EU regulatory changes. Covers history, ethics, and patient autonomy.

An in-depth comparison of the FDA and EMA, updated for 2026. Learn the key differences in US and EU drug approval processes, review timelines, GMP, FDORA reforms, EU pharma legislation overhaul, and regulatory frameworks.

A comprehensive guide to the clinical trial protocol, updated for 2026 with ICH E6(R3) and ICH M11 developments. Learn its core components, importance in research, and how to manage complexity and amendments for successful trials.

Learn why the drug development timeline averages 10-15 years. Updated for 2026 with AI drug discovery milestones, ICH E6(R3), FDA AI guidance, and latest cost and success rate data. Covers every stage from discovery through approval.

Learn about key clinical trial acronyms. This guide explains the definitions, history, and roles of GCP, ICH, IRB, EDC, and eTMF in clinical research. Updated with ICH E6(R3) finalization (2025), FDA DCT guidance, and current market data.

Learn the key clinical research roles. Updated for 2026 with ICH E6(R3), this guide defines the responsibilities of the PI, CRC, CRA, and Clinical Project Manager in clinical trials

Learn the critical differences between preclinical and clinical research in drug development, from lab-based toxicology (GLP) to human clinical trials (GCP). Updated with FDA Modernization Act 3.0, NAMs guidance (2026), AI drug discovery trends, and current attrition data.

Compare full-service (FSO) and Functional Service Provider (FSP) CRO outsourcing models. Updated for 2026 with market data, ICH E6(R3), BIOSECURE Act, AI trends, and hybrid model adoption

Explore the distinct roles in clinical trials. Learn the specific responsibilities of sponsors, CROs, and sites, from protocol design to regulatory compliance u

Learn what a Contract Research Organization (CRO) is and its critical role in modern drug development. Updated for 2026 with latest market data, ICH E6(R3) guidelines, BIOSECURE Act impacts, AI adoption trends, and major M&A activity including Thermo Fisher/Clario and Syneos Health privatization.

An in-depth guide to the four phases of clinical trials, updated for 2026. Learn Phase I-IV objectives, FDA single-trial approval policy, ICH E6(R3) updates, AI-driven trial design, and current success rates

Learn the complete data cleaning process in clinical trials. This guide covers clinical data management (CDM) best practices, error detection, and regulatory co

A complete guide to clinical trial site close-out, updated for 2026. Covers data integrity, IP accountability, archiving, and regulatory compliance per ICH E6(R3), EU CTR, UK 25-year retention, and FDA safety reporting guidance

Learn the technical requirements for GLP audit trails (updated April 2026). Covers compliant file systems, WORM storage, EU Annex 11 revision, OECD IT Security guidance, and network shares for data integrity

Need GLP compliance software? Our 2026-updated guide reviews the top 5 systems for data integrity, 21 CFR Part 11, and audit trails. Compare MasterControl, Veeva Vault, Honeywell TrackWise, LabWare LIMS, and Qualio with the latest AI and regulatory developments.

Learn about 21 CFR Part 58, the FDA's Good Laboratory Practice (GLP) regulations for nonclinical studies. Updated for 2026 with recent enforcement actions, OECD IT security guidance, and New Approach Methodologies (NAMs)

A technical guide to creating a GLP-compliant audit trail on macOS (Sequoia 15 / Tahoe 26). Learn to meet data integrity rules (21 CFR Part 11, FDA CSA 2025) using APFS, FSEvents & Endpoint Security

Explore the FDA Pre-license Inspection (PLI) process for biologics. This guide explains the PLI's purpose, scope, cGMP requirements for BLA approval, and the latest 2025-2026 developments including AI-driven inspection targeting, remote assessments, and the BIOSECURE Act.

An educational review of top FDA compliance monitoring companies (updated March 2026). Compare leading QMS software platforms and expert regulatory consulting firms for pharma & medical devices, including QMSR/ISO 13485 transition guidance.

An overview of glial cells (neuroglia), the essential non-neuronal cells in the brain. Learn the functions of astrocytes, oligodendrocytes, microglia, and more, including 2025-2026 breakthroughs in astroengrams, microglia replacement therapy, and remyelination.

Explore the state of Real-World Evidence (RWE) analysis in 2025-2026. This guide covers RWD sources, analytical methods, FDA/EMA regulatory frameworks, ICH E6(R3), synthetic control arms, AI-driven RWE platforms, and compares RWE with RCTs

Updated for 2026: This guide analyzes 10 leading patient recruitment services and software platforms — including Tempus AI, TriNetX, and Clariness — used to accelerate clinical trial enrollment

An educational guide to leading pharmacovigilance service providers (updated April 2026). Learn about the PV outsourcing market, regulatory drivers including FDA AEMS and EU AI Act, and compare top drug safety CROs including Fortrea, IQVIA, ICON, and more

Updated 2026 guide to biomarker testing coordination services in oncology. Covers navigator roles, FDA companion diagnostic reclassification, liquid biopsy adoption, NCCN guideline updates, and AI-driven diagnostics for precision medicine.

Learn about the medical writing and scientific publication planning industry. This guide covers market size, key services like regulatory writing, GPP 2022 guidelines, AI tools, and leading service providers

Learn the fundamentals of KOL research and mapping in 2026. This guide covers AI-driven methods for KOL identification, key data sources, network analysis, and emerging platforms in healthcare and life sciences.

A comprehensive guide to MLR review software for pharma and life sciences (updated March 2026). Compare the top platforms including Veeva AI Agents, EVERSANA ORCHESTRATE MLR, and more.

Explore the 2025 rare disease landscape, affecting 300 million people globally. Learn about diagnostic challenges, orphan drug development, and future policy di

Learn about the ICH Q8 guideline for pharmaceutical development. This in-depth guide explains Quality by Design (QbD), QTPP, CQAs, design space, and includes 2025-2026 updates on ICH Q13, Q14, and AI in manufacturing.

Explore a comprehensive list of the top pharma news websites and biotech publications in 2026. Learn about sources for drug development, regulatory changes, patent cliff coverage, and market intelligence.

An educational analysis of the 2026 drug patent cliff. View a complete list of patent expirations, including Ozempic & Januvia, and their impact on generic drug

Explore open source PHI de-identification solutions for clinical data. This 2026-updated guide covers rule-based, ML, LLM-based, and hybrid tools for scrubbing text per HIPAA standards

Side-by-side comparison of all 5 ChatGPT plans with pricing, features, and limits. Find which plan fits your needs: Free, Plus, Pro, Business, or Enterprise.

A comprehensive 2026 guide to ICH Q7, Q8, & Q9. Learn the core principles of GMP for APIs (Q7), Quality by Design/QbD (Q8), and Quality Risk Management/QRM (Q9)

Learn about CDISC standards for clinical trial data. This guide explains SDTM, ADaM, CDASH, and Define-XML with concrete examples for regulatory submissions.

Understand the FDA 510(k) premarket notification process for Class II medical devices. Updated for 2026 with QMSR, AI/ML device frameworks, MDUFA VI, and current clearance data.

Explore how AI impacts Good Documentation Practice (GDocP) and ALCOA+ principles in life sciences. Updated with the 2026 joint FDA-EMA 10 Guiding Principles, EU AI Act implementation timeline, and the EMA's first AI qualification opinion. Learn about efficiency gains, data integrity risks, and regulatory compliance strategies.

Explore why AI in radiology assists, but doesn't replace, human radiologists. Learn about the limits of AI and the critical role of clinical context and experti

Learn to build a Model Context Protocol (MCP) server for Power BI. Updated for 2026 with OAuth 2.1 auth, Microsoft's official Power BI MCP servers, and Entra ID integration.

Learn about the most in-demand pharma roles for 2025-2026. This analysis covers top pharmaceutical jobs in AI, advanced manufacturing, and clinical development, including $370B+ in US reshoring investments and workforce restructuring trends.

Learn about ISO 13485:2016, the QMS standard for medical devices. Updated for 2026 with FDA QMSR effective date, EU MDR/EUDAMED timelines, AI/ML guidance, and certification data

Is ISO 9001 still used in biotech? Updated for 2026 with ISO 9001:2026 revision details and ICH E6(R3). Analyzes how the QMS standard supports regulatory compliance, complements GMP, and improves quality in pharma & life sciences

Learn what ISO/IEC 17025 is in this complete guide. Explore requirements for testing and calibration laboratory accreditation, including the new 2025 edition changes, transition timeline, and key benefits

An in-depth technical analysis of Kimi K2 and K2.5, the trillion-parameter MoE LLMs from Moonshot AI. Covers architecture, training innovations, benchmark results, and the K2.5 successor with agent swarm capabilities

Explore the updated ICH E6(R3) Good Clinical Practice (GCP) guideline. Learn about the key changes from R2, the new structure with Annex 1 & 2, and risk managem

An analysis of leading pharmaceutical consulting firms in 2026, from global strategy to specialized AI boutiques. Review key services, $30B+ market data, AI trends, and firm rankings.

Learn what a CRDMO is and how this integrated model combines CRO and CDMO services. Updated for 2026 with BIOSECURE Act impact, latest market data, and industry M&A

Updated 2026 guide to pharmaceutical compliance software for GxP and QMS. Compare Veeva, MasterControl, ETQ & more with FDA QMSR, CSA guidance, and Gartner MQ coverage

Analysis of the top pharmaceutical packaging companies in a $116B+ market (2025). Covers the Amcor-Berry merger, BD's GLP-1 investments, Gerresheimer developments, and innovations in prefilled syringes and sustainable packaging.

A guide to biotech sales meeting booking for life sciences. Learn data-driven best practices for appointment setting, from prospect research to personalized outreach and AI-powered prospecting tools

Learn how to ethically source U.S. HCP contact information, including email and mobile numbers. Updated for 2026 with new TCPA consent rules, 19 state privacy laws, and FTC enforcement trends.

A detailed 2025 comparison of ChatGPT Enterprise vs. Microsoft Copilot. Learn the key differences in features, integration, security, and enterprise AI strategy

Learn why high-cost specialty drugs for chronic diseases are often mail-order only. Updated for 2026 with FTC findings, PBM reform, biosimilar trends, and the latest distribution network data.

Updated 2026 guide to US mail-order specialty pharmacies. Covers $751B market, top players (CVS, Optum, Express Scripts), FTC PBM reform, Walgreens-Sycamore split, GLP-1 growth, and specialty drug distribution trends.

Learn what specialty pharmacy is, how it differs from retail, and its role in managing high-cost specialty medications. Updated for 2026 with the latest on GLP-1 drugs, FTC PBM reforms, IRA out-of-pocket caps, and gene therapy growth.

Explore the electronic prior authorization (ePA) workflow for GLP-1 drugs. Updated for 2026 with CMS-0057-F rule, Medicare GLP-1 Bridge program, MFN pricing deals, oral Wegovy approval, and AHIP reform pledges.

Learn about OCR and Document AI performance on real pharma documents. This 2026-updated report analyzes accuracy benchmarks for data extraction from clinical trials and SOPs, covering VLMs, agentic AI, and FDA CSA guidance

Learn about Veeva MyInsights development, its evolution to Vault CRM X-Pages with AI Agents (2025-2026), key timelines, technical architecture, example dashboards, and the Salesforce migration deadline. Updated March 2026.

Learn how open source software like R and Python is changing pharma R&D. From Novo Nordisk's first R-based FDA submission to AlphaFold 3's open-sourcing, we analyze the accelerating shift from proprietary systems to collaborative drug discovery models.

Explore US hospital readmission rates by state with 2025-2026 data. This report analyzes 30-day readmission statistics, factors driving state variation, FY 2026 HRRP penalty updates, and CMS policy changes including Medicare Advantage integration

Learn evidence-based strategies to reduce hospital readmission rates. Updated for 2026 with HRRP penalty changes, the TEAM bundled-payment model, ML-driven risk prediction advances, and CMS Accountable Health Communities results.

Learn the complete KOL/HCP mapping process for pharma. This playbook covers data-driven methods for KOL identification, segmentation, and network analysis.

Learn about clinical data interoperability with this updated guide to HL7 FHIR (R5/R6), eSource, and ePRO. Covers ICH E6(R3), FDA FHIR initiatives, standards, use cases, and challenges in modern research.

Updated 2026 guide to pharmacovigilance technology covering GenAI/LLMs in PV, AI-powered case processing, signal detection, FDA-EMA AI guidance, and data standards for drug safety
Learn about the FDA's AI/ML medical device tracker. With 1,451 devices authorized through 2025 and 295 cleared in 2025 alone, we analyze authorization trends, specialty breakdown, foundation model milestones, and the evolving regulatory framework

What is a TPA? Learn the role of Third-Party Administrators in self-funded health insurance, from claims processing and plan administration to cost containment. Updated with 2026 regulatory reforms including the CAA 2026 and DOL transparency rules.

An in-depth analysis of the Swiss healthcare system (updated March 2026). Covers the EFAS reform, TARDOC tariff, EPR overhaul, rising premiums, and the unique decentralized model of mandatory insurance (LAMal/KVG).

McKesson, Cencora, and Cardinal Health control over 90% of US drug distribution ($900B+ combined revenue). Full market analysis with FY2025 financials and 2025-2026 developments.

Explore the top 3 PBMs controlling 80% of the U.S. market. Updated with 2026 federal reform law, FTC Express Scripts settlement, DOJ UnitedHealth probe, and landmark rebate pass-through requirements.

An in-depth analysis of the leaders in pharma digital transformation, examining the strategies of companies like AstraZeneca, Roche, Pfizer, Eli Lilly, and Sanofi with AI, generative AI, and cloud technologies — updated with 2025-2026 developments including Lilly's $1B NVIDIA AI lab and FDA AI guidance.

Get a detailed guide to Oracle Life Sciences solutions. Learn about key products for clinical trials (Clinical One 26.1), pharmacovigilance (Argus Safety), Health Data Intelligence, and AI-powered analytics.

An in-depth guide to Veeva Vault. Learn how this cloud platform for life sciences manages regulated content, ensures GxP compliance, and unifies data management

A research-based guide to healthcare chatbot platforms (updated March 2026). Compare AI solutions for patient engagement, symptom triage, and HIPAA compliance including Hippocratic AI, Ada Health, Microsoft Healthcare Agent Service, and more.

Learn key principles for custom pharmaceutical software app design. Updated for 2026 with FDA CSA guidance, GAMP 5 Second Edition, GxP compliance, 21 CFR Part 11, and AI/ML trends

Learn about SMART on FHIR for EHR interoperability. This guide covers the FHIR Coverage resource, CDS Hooks, TEFCA, Da Vinci IGs, and the latest 2025-2026 regulatory updates for building modern healthcare applications.

Learn the essential HIPAA compliance requirements for healthcare startups. This updated 2026 guide compares SOC 2 and HITRUST to help you choose the right framework for PHI protection, with the latest breach statistics, enforcement actions, and regulatory developments.

Updated 2026 guide to Medicare Part D vs. Medicare Advantage drug plans, including IRA drug price negotiations, $2,000 OOP cap, and Medicaid managed care changes under the 2025 reconciliation law.

Learn how the NCPDP SCRIPT standard enables electronic prior authorization (ePA). Explore the technical workflow, Surescripts CompletEPA, CMS-0057-F requirements, AI automation, and FHIR-based PA APIs shaping 2026-2028 compliance.

Learn the key differences between NDC and RxNorm drug coding systems. Updated for 2026 with FDA's finalized 12-digit NDC rule, HCPCS J-code updates, and RxNorm developments.

Learn what Pharmacy Benefit Managers (PBMs) are and their role in the U.S. healthcare system. Updated for 2026 with FTC Express Scripts settlement, CAA 2026 federal PBM reform, Medicare Part D changes, and latest state regulations.

Learn about X12 EDI integration for startups (updated March 2026). This guide compares top vendors and solutions, from traditional VANs to modern cloud EDI-as-a-Service platforms with AI-powered capabilities.

Learn the key differences between pharmacy vs. medical benefit and how this structure impacts insurance coverage for GLP-1 drugs for diabetes and obesity, including the 2025–2026 Medicare BALANCE Model, manufacturer price cuts, and employer coverage trends.

Learn what drug formulary tiers are and how they affect prescription costs. Updated for 2026 with CMS-0057-F ePA rules, gold card laws, and IRA drug cost caps.

Learn how PBMs work and compare self-funded vs. fully insured health plans. Updated for 2026 with FTC settlement, federal rebate pass-through mandate, and latest KFF data on financing models, ERISA, risk, and PBM impact on drug costs.

A deep-dive tutorial on the EDI X12 270/271 transaction sets. Learn how this HIPAA standard enables real-time pharmacy benefit and eligibility verification, updated with 2025 Surescripts data, CMS-0057-F FHIR mandates, and NCPDP RTPB v13 requirements.

Compare the top 5 Availity alternatives for healthcare claims management in 2026. Covers Waystar (post-IPO), TriZetto (data breach impact), Experian Health, Office Ally, and SSI Group with updated KLAS rankings and market data.

Learn the technical structure of the ANSI ASC X12 EDI format. This guide explains B2B data exchange via envelopes (ISA/IEA), segments, data elements, and delimi

An educational analysis of Nirvana Health's history, its evolution from PBM RxAdvance, and its Aria platform for automating payer and PBM operations with AI, RPA, and the 2025 Transaction-as-a-Service (TaaS) model

A 2025 overview of AI in radiology, covering FDA approvals, clinical adoption rates, and key technologies from CNNs to foundation models for medical imaging.

Learn about the X12 EDI standard for electronic data interchange. Updated for 2026, this guide covers X12 Version 008060, common transaction sets (850, 837), HIPAA updates, EDI-API hybrid integration, and the $41B+ EDI market.

Learn the fundamentals of EDI mapping for CPG suppliers. This 2026-updated guide covers EDI standards (ANSI X12, EDIFACT), AI-driven mapping automation, hybrid EDI+API integration, and retailer compliance strategies.

Learn about Cohere Health, the health-tech firm using AI to automate prior authorization. Updated for 2026 with ZignaAI acquisition, record growth, and expanded platform coverage

Explore the difference between autonomous AI agents and structured AI workflows. Learn why deterministic AI pipelines dominate production deployments in 2025.

Learn how IBM Quantum is applied in pharmaceutical R&D. This guide covers molecular simulation, hybrid workflows, the Nighthawk and Starling processors, and key partnerships with Moderna, Algorithmiq, and Cleveland Clinic.

Learn how the Availity platform serves as an EDI gateway for payers. This 2026-updated analysis covers its FHIR solutions, Rapid Recovery cybersecurity, AI-powered prior authorization, and CMS-0057 compliance.

With the rise of vibe coding, many question Agile's future. Our analysis shows Agile remains crucial for managing risks and ensuring quality in AI-driven projec

Learn what a semantic layer is. This technical guide explains how it bridges data sources and BI tools, creating unified metrics and business-friendly data mode

A detailed 2025 analysis of the GxP managed services market for pharma & life sciences. Learn about trends, regulatory drivers, and GxP compliance challenges.

An in-depth analysis of the specialty pharmacy industry covering key players (PBMs, distributors), market trends, biosimilar competition, PBM reform, and the economics of high-cost specialty drugs. Updated March 2026.

Learn about the ANSI X12 EDI protocol. This guide explains the structure of X12 transaction sets, with a deep dive into healthcare's 270/271 and 278 transactions, including CMS-0057-F prior authorization mandates and the X12-to-FHIR transition

Learn why over 85% of clinical trials face delays. Updated for 2026, this analysis covers key scientific, operational, and regulatory challenges from Phase I to III, including site activation bottlenecks, AI-driven recruitment, and ICH E6(R3) GCP updates.

Learn the complete history of GLP-1 drugs from the incretin effect to 2026 breakthroughs. Covers exenatide, semaglutide, tirzepatide, oral Wegovy, retatrutide, CagriSema, and the evolution from diabetes to obesity treatment.

An in-depth guide to US drug formularies, pharmacy benefits, and prior authorization (PA). Learn how P&T committees, tiers, ePA systems, and the CMS-0057-F interoperability rule shape drug coverage and costs

Get a complete list of Real-Time Benefits Check (RTBC) modules and providers. Learn how solutions from Surescripts, DrFirst, Arrive Health (formerly RxRevu), GoodRx & more work within EHRs. Updated for 2026.

Learn what a health insurance formulary is. This guide explains the drug list, tiers, and cost-sharing used by PBMs to control prescription costs and access.

An evidence-based analysis of the top 5 electronic prior authorization (ePA) platforms. Compare CoverMyMeds, Surescripts, OptimizeRx, DrFirst & Availity — updated with 2025-2026 CMS-0057-F milestones, Touchless PA, and AI-driven workflows.

Learn about the NCPDP SCRIPT standard, the U.S. foundation for electronic prescribing (e-prescribing). Updated for 2026 with HTI-4 Final Rule, v2023011 migration, and latest Surescripts data.

A technical analysis of cost-effective LLMs (updated March 2026). Compare API pricing, performance benchmarks, and features for Gemini 3.1 Flash-Lite, Claude Haiku 4.5, GPT-5 Mini, Grok 4.1 Fast, DeepSeek V3.2, and more.

Learn the 9 ALCOA+ principles for GxP data integrity. Updated for 2026 with ICH E6(R3) finalization, EU GMP Chapter 4 ALCOA++ draft, and latest FDA enforcement trends

Explore Veeva QualityOne, the unified cloud QMS for regulated industries like CPG & chemicals. Updated for 2026 with AI Agents, LIMS, new modules, and customer case studies.

A complete LLM API pricing comparison for 2025. Analyze token-based costs for OpenAI (GPT-5), Google Gemini, Anthropic Claude, Grok, and DeepSeek models.

An educational analysis of AI biotech funding in 2025. Explore venture capital investment trends, funding data, and key deals in AI for drug discovery.

Explore the key drivers behind major pharma and CRO layoffs in 2025-2026. This analysis covers economic pressures, patent cliffs, and R&D shifts at top companie

Analysis of AI medical devices in 2025. Learn about market growth, 950+ FDA-cleared devices, new regulations like the EU AI Act, and key clinical challenges.

Learn what an electronic Investigator Site File (eISF) is and its role in clinical trials. Updated for 2026 with ICH E6(R3) finalization, latest adoption data, and eISF vs. eTMF comparison

Comprehensive guide to Regulatory Information Management (RIM) systems and ISO IDMP standards, covering EMA PMS deadlines, eCTD v4.0 timelines, AI-powered RIM tools, and compliance strategies for life sciences organizations (updated March 2026).

Learn about the TMF Reference Model (now TMF Standard Model under CDISC), the industry-standard taxonomy for organizing Trial Master File documents in clinical trials. Covers TMF SM v1, ICH E6(R3) alignment, and AI-powered eTMF automation.

Updated 2026 guide to finding drugs in the clinical pipeline (Phase I-III). Learn to use ClinicalTrials.gov (550K+ studies), CTIS, commercial databases, and AI-powered tools like Pharmaprojects+ and TuneLab.

Prepare for your life sciences role with our updated 2026 guide to top Veeva Vault interview questions. Covers Vault CRM migration, AI Agents, platform features, and compliance topics

Explore our 2025 analysis of the top 25 pharmaceutical companies by AI patent filings. Learn who leads in AI R&D, key technology areas, and strategic trends.

Learn to design a ChatGPT workshop for biotech professionals. Updated for GPT-5 and 2026 regulatory frameworks, this guide covers LLM fundamentals, practical use cases, and prompt engineering for life sciences.

Updated 2026 framework for validating generative AI in GxP systems. Covers 21 CFR Part 11, EU Annex 22, ISPE GAMP AI Guide, FDA CSA guidance, and FDA-EMA joint AI principles

Explore a technical blueprint for an RWE platform architecture, detailing how to integrate real-world data (RWD) from EMR, claims, and genomics sources. Updated with 2025-2026 developments including EHDS regulation, DARWIN EU progress, and FHIR R5.

Learn about Japan's drug approval process, updated for 2026. This guide covers PMDA & MHLW roles, review timelines, drug lag, the 2025 PMD Act amendments, eCTD v4.0 mandate, and expedited pathways like SAKIGAKE.

Explore the top MCP servers for biotech. Learn how the Model Context Protocol connects AI agents and LLMs to critical databases for genomics and drug discovery.

Learn the key differences between Anthropic's Claude Skills and the Model Context Protocol (MCP). This guide explains their architecture and use cases.

Explore how AI in remote patient monitoring (RPM) improves clinical outcomes. Updated for 2026 with FDA PCCP guidance, Apple Watch hypertension clearance, CMS reimbursement changes, and market projections.

Learn about FutureHouse, the nonprofit AI research lab. This guide covers its platform, AI agents (Crow, Falcon, Owl, Phoenix, Finch), the Robin discovery system, ether0 reasoning model, and Edison Scientific spinout.

An in-depth analysis of the 2025 Computer System Validation (CSV) job market. Learn about validation specialist salaries, key skills, and career trends.

An in-depth analysis of 1,000+ medical device companies in the SF Bay Area, covering the medtech ecosystem, key players like Intuitive Surgical ($10B+ revenue), Penumbra ($14.5B acquisition), and CeriBell's IPO, plus VC funding and market trends through 2026.

Updated guide to Sora 2, OpenAI's AI video model and TikTok-style app. Covers the Disney $1B deal, pricing changes, Android launch, download decline, and competition from Google Veo 3.

An educational analysis of California's AB 1415 and SB 351. Learn how these laws impact private equity and VC in healthcare, including OHCA review and new rules

Learn about Humanity's Last Exam (HLE), the Nature-published AI benchmark testing true LLM reasoning with 2,500 expert-level questions. Updated with 2026 leaderboard scores from GPT-5, Claude Opus, and Gemini 3.
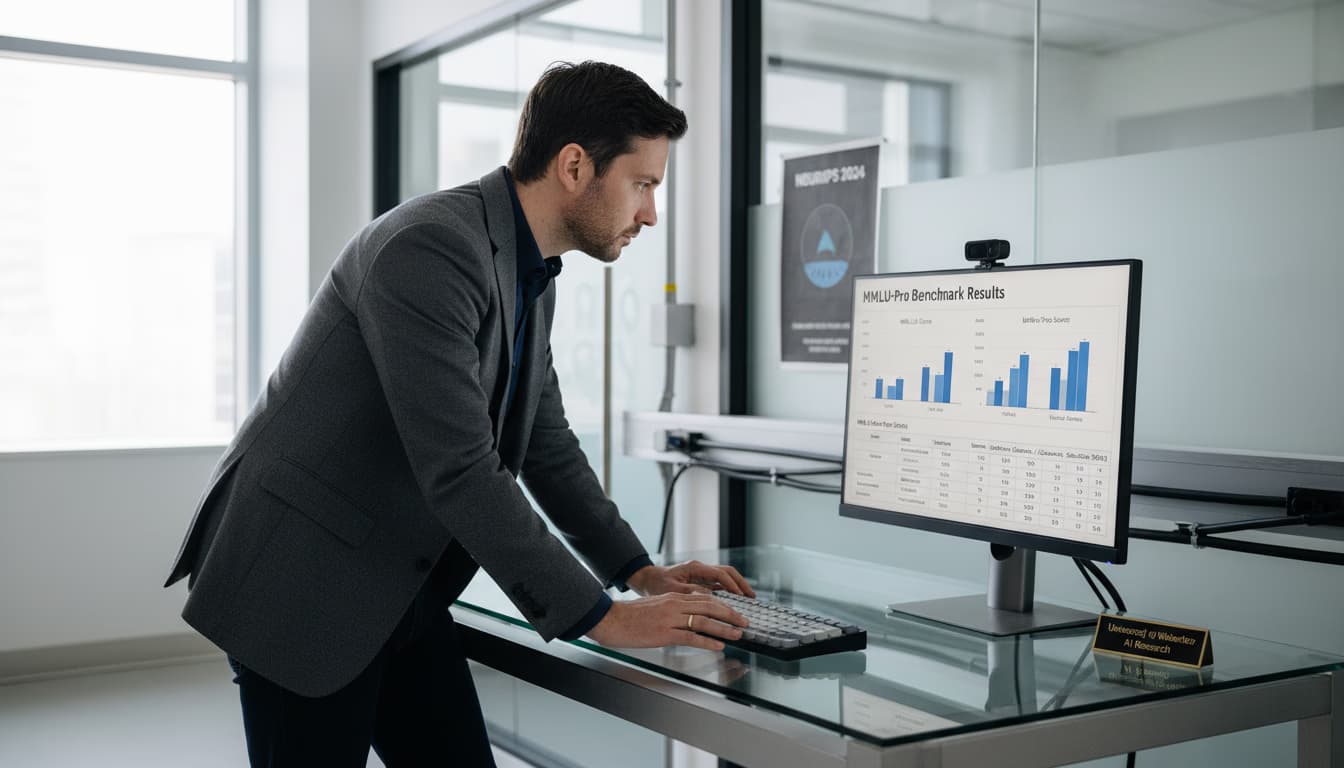
Learn about MMLU-Pro, the advanced AI benchmark designed to overcome MMLU's limitations. This guide explains its design, dataset, and impact on LLM evaluation.

NVIDIA H100 GPU rental rates from $1.49/hr (Vast.ai) to $6.98/hr (Azure). Compare AWS, GCP, Lambda, RunPod, CoreWeave and more.

Updated 2026 comparison of NVIDIA data center GPUs: Blackwell Ultra B300, B200, GB200 NVL72, H100, H200, A100 & L40S — specs, FLOPS, NVLink, cloud availability & Vera Rubin roadmap.

Explore the exponential rise in AI compute demand in biotech. This 2025 report analyzes key statistics, infrastructure needs, and trends in drug discovery and g

Explore the AIME 2025 benchmark, a key test for AI mathematical reasoning. See how models like GPT-5 score over 94% and compare LLM performance on Olympiad-leve

Learn why DeepSeek's AI inference is up to 50x cheaper than competitors. This analysis covers its Mixture-of-Experts (MoE) architecture and pricing strategy.
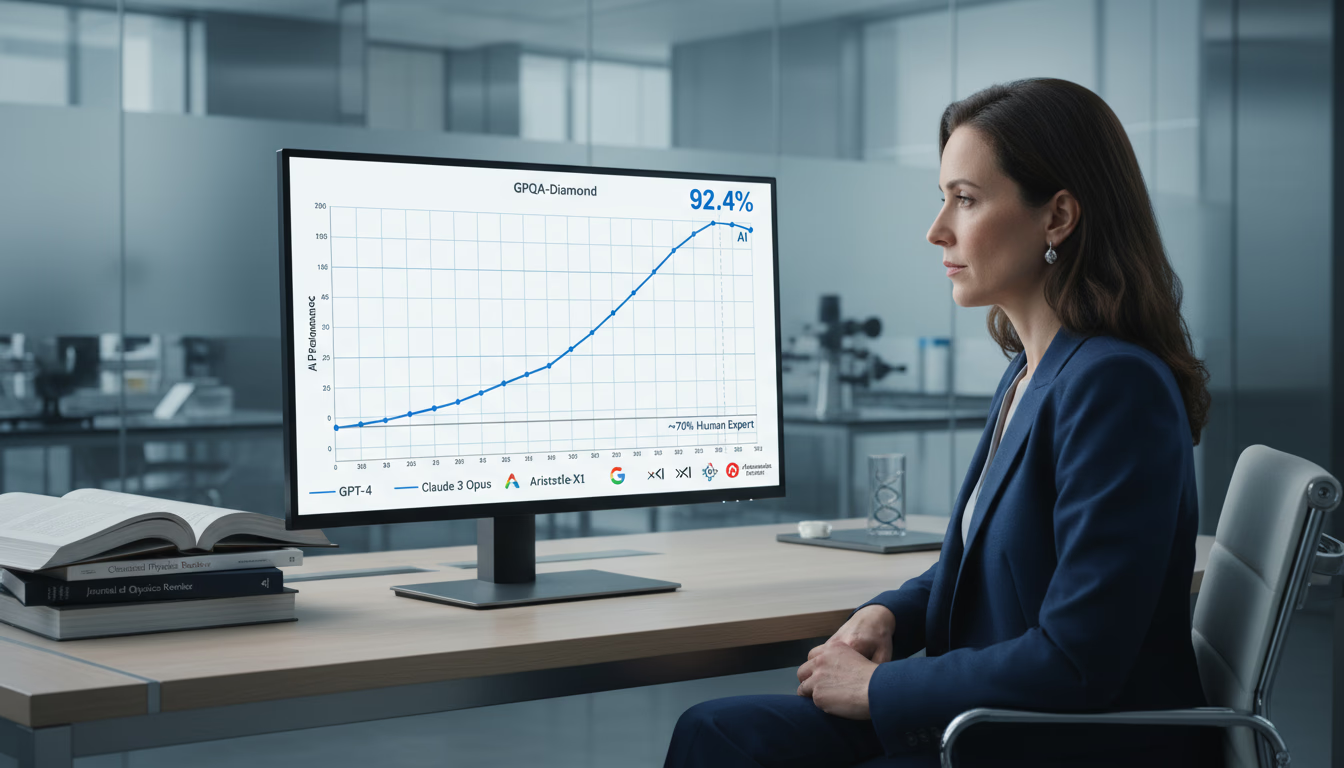
GPQA-Diamond scores updated through 2026: Gemini 3.1 Pro (94.1%), GPT-5.2, Claude Opus 4.6, Aristotle-X1, and more. See which AI models beat PhD experts on 198 graduate-level science questions.

A technical guide to building a HIPAA-compliant OCR pipeline for healthcare. Learn key security controls, PHI handling, encryption, and cloud architecture.

An educational guide to HPC in life sciences. We review top lab IT specialists and solutions for genomics, drug discovery, and bioinformatics data analysis.

Updated 2026: Learn what NVIDIA BioNeMo is and how it accelerates drug discovery. This guide explains its components, latest models (RNAPro, ReaSyn v2), the $1B Lilly partnership, and deployment for generative AI in biopharma.

Learn the strict physical requirements for deploying NVIDIA HGX platforms from Hopper to Blackwell. Covers power (10-140 kW/rack), liquid cooling, rack design, and infrastructure for DGX H100, B200, and GB200 NVL72 systems.

Analyze the leading data center providers for private AI solutions in 2025-2026. This guide compares on-prem and hybrid infrastructure from AWS, Azure, HPE, Dell, Cisco, VMware/Broadcom, Equinix, and NVIDIA, including Blackwell GPUs and sovereign cloud trends

An in-depth analysis of ChatGPT Atlas, OpenAI's AI browser — updated March 2026 with latest feature updates, 900M+ ChatGPT users, Google Chrome's Gemini response, and the evolving AI browser war.

Get a complete 2025 analysis of Clinical Trial Management System (CTMS) software. This guide compares top vendors, core features, and eClinical market trends.

An analysis of how Docusign (formerly DocuSign) is used for electronic signatures in pharma and life sciences. Learn how it can meet FDA 21 CFR Part 11 compliance with its Life Sciences modules and IAM platform, updated for 2026.

Learn about Lorenz docuBridge, a key eCTD publishing software for biotech. Updated for 2026 with v25.2 features, eCTD v4.0 EU support, verifAI, and 2,000+ installations

A comprehensive review of the NVIDIA DGX Spark (updated March 2026). Explore its specs, 2.5x post-launch performance gains, $4,699 pricing, and how it compares to AMD Strix Halo and Apple M4 Ultra for local AI development

Learn about Dassault Systèmes' QUMAS EDMS for life sciences. This guide covers its features, EDMS 2026 release, AI integration, and use cases for GxP and 21 CFR Part 11 compliance.

An educational analysis of the remote inspection software market for life sciences. Learn about market size, growth drivers, QMS vendors, and PSC's ACE Inspecti

An in-depth 2025 analysis of AI accelerators from Cerebras, SambaNova, and Groq. Compare their unique chip architectures, funding, and performance for AI worklo

An in-depth analysis of the top 10 open source chatbot platforms for local deployment in 2026. Compare features, adoption data, and use cases for Rasa (CALM), Botpress, Microsoft Copilot Studio, and more.

Compare on-prem AI infrastructure from Dell, HPE, Lenovo, Supermicro & Cisco. Analyze NVIDIA GB200/GB300 NVL72 and Blackwell Ultra hardware specs, cooling, software, and performance. Updated February 2026 with VCF 9.0, Vera Rubin roadmap, and latest vendor announcements.

Learn about NVIDIA NVLink, the high-speed GPU interconnect from NVLink 1.0 through NVLink 6.0 (Rubin). This guide covers architecture, bandwidth specs, NVSwitch, NVLink Fusion, and comparisons with AMD Infinity Fabric, CXL, and UALink.

Learn about Innodisk APEX AI servers for running local AI models. Updated for 2026 with Dragonwing series, IEC 62443 security certification, and latest edge AI market data.

An enterprise guide to LLM inference hardware in 2026. Compare NVIDIA Blackwell/Rubin, AMD MI350X, Cerebras, SambaNova SN50, and other AI accelerators for running powerful LLMs on-premises.

An in-depth analysis of the HMMT25 AI benchmark for testing advanced mathematical reasoning in LLMs. See how models like Grok-4, GPT-5, and Gemini 3 perform on complex contest math problems, and how newer benchmarks like FrontierMath are raising the bar.

Explore DeepSeek-OCR, an AI system that uses optical compression to process long documents. Learn how its vision-based approach solves long-context limits in LL

Explore the Nvidia Blackwell vs Hopper GPU architectures. Key technical differences in tensor cores (FP4/FP6), memory, MLPerf benchmarks, and the road to Rubin — updated for 2026.
Explore a data-driven comparison of the AI boom and the dot-com bubble. We analyze key metrics like valuations, profitability, and VC funding — updated with 2026 data including NVIDIA's $4.3T market cap, OpenAI's $730B valuation, and $258.7B in AI VC funding.
Explore the key differences between AI engineers and software engineers in 2026. Compare skills, salaries, responsibilities, and career growth with the latest LinkedIn and industry data.

Compare Zapier vs n8n for AI workflow automation. This in-depth report covers key differences in pricing, integrations, self-hosting, and AI capabilities.

Learn what context engineering is and how it improves AI and LLM reliability. Updated for 2026, this guide covers RAG, MCP, agentic AI, and why context engineering has replaced prompt engineering
With 40% of agentic AI projects facing cancellation, learn why durable orchestration with Temporal.io is vital for reliable multi-agent workflows in production

Get a detailed 2025 salary guide for a Medical Data Annotation Specialist. Learn about compensation in pharma & biotech, including US and UK pay benchmarks.

An in-depth guide to pharmaceutical MES and MOM software (updated 2026). Compare top vendors including PAS-X, Opcenter, and PharmaSuite 12.00, understand cGMP compliance, and learn how cloud-native MES enables electronic batch records

Learn how Reinforcement Learning from AI Feedback (RLAIF) reduces medical AI annotation costs. This guide covers the RLAIF method, its benefits over RLHF, and u

Explore the technical architecture of RLHF for drug discovery. Learn how reward models and policy optimization align generative AI with expert chemist feedback, with 2025-2026 clinical validation data.

Build a safe and reliable clinical LLM using an RLHF pipeline. This guide covers the architecture, SFT, reward modeling, DPO, GRPO, and AI alignment for healthcare, updated with 2025-2026 developments including Med-Gemini and FDA guidance.

An in-depth analysis of RLHF platforms for biotech (updated Feb 2026). Compare Scale AI (post-Meta deal), Labelbox, Appen, Surge AI, and in-house solutions on capabilities, cost, and HIPAA compliance.

Explore the 2025 market for medical data labeling. This guide covers market size, growth, and the regulatory landscape, including HIPAA and the new EU AI Act.

Explore the digital therapeutics (DTx) market. This analysis covers market size, growth trends, key drivers, regulatory frameworks, and future outlook for 2025.

Learn about the EU AI Act's impact on pharma. Updated for 2026 with the Digital Omnibus proposal, GPAI Code of Practice, and revised Product Liability Directive. Includes risk classification, compliance steps, flowchart & SOP starter kit.

Explore 2025 data on AI adoption in U.S. hospitals. This report covers key statistics, use cases like sepsis detection, EHR integration, and adoption disparitie

A 2026-updated technical comparison of Databricks vs. Snowflake for life sciences. Explore the lakehouse and AI data cloud for genomics, clinical data, Mosaic AI, Cortex AI, and ML workloads.

An analysis of GLM-4.6 and its successor GLM-5, the leading open-source coding models. Compare benchmarks against Claude Opus 4.6, Sonnet 4.6, and GPT-5.3-Codex, plus hardware requirements

An in-depth 2025 analysis of the leading medical imaging and digital pathology AI vendors. Covers market size, deep learning trends, and top company profiles.

Our 2025 report analyzes the critical AI skills gap in the pharmaceutical industry, detailing the talent shortage with data and exploring key solutions.

An in-depth guide to IT system assessment in pharma M&A due diligence. Covers infrastructure, data, cybersecurity, AI governance, NIS2/CRA compliance, and FDA cybersecurity guidance for 2025-2026 deals.

Explore a detailed cost-benefit analysis of RTSM implementation in clinical trials. Learn how RTSM systems reduce drug waste by 15-30%, save millions, and align with ICH E6(R3) and AI-driven forecasting trends in 2025-2026.

Learn how target trial emulation provides a structured framework for drawing causal inference from real-world evidence (RWE), with 2025 updates on the TARGET reporting guideline, ICH M14, and FDA guidance.

Explore biotech salary trends across global regions, updated with 2024-2025 data. Compares pay in top hubs like the US, Switzerland, and Asia-Pacific with cost-of-living analysis and funding recovery trends.

Updated 2026 Epic vs Cerner AI comparison. Native AI Charting, CoMET foundation models, Oracle's next-gen EHR, Clinical AI Agent results, and the reshaping ambient scribe market.

Learn the GAMP 5 software categories for computerized system validation. This guide explains each category with examples, risk levels, and how they align with the 2025 ISPE GAMP AI Guide and FDA CSA framework.

A comprehensive guide to lab equipment qualification (IQ/OQ/PQ). Learn the process, regulatory standards including FDA's finalized CSA guidance, and how AI-powered automation streamlines validation and compliance.

An in-depth review of Certara's drug development software suite for MIDD, updated for 2026. Covers Phoenix WinNonlin, Phoenix Cloud, the Simcyp PBPK Simulator, Certara IQ AI-powered QSP platform, and ICH M15 adoption.
Learn the key differences between GAMP 4 and GAMP 5. Updated for 2026 with FDA's finalized CSA guidance, ISPE GAMP AI Guide, and EU GMP Annex 11/22 drafts. Clear migration path for pharma IT.

Learn about Japan's PMDA regulatory pathways for drug approval under the PMD Act, including 2025 amendments. Covers standard and expedited options like Sakigake, Orphan Drug, and Conditional Approval

Updated 2026 guide to ICH M7 mutagenic impurity assessment software including Derek Nexus 6.5, Sarah Nexus 5.1, Leadscope 2025.0, OECD QSAR Toolbox 4.8, AmesNet deep learning, and nitrosamine regulatory developments.

A detailed analysis of the global pharmaceutical market, forecast to reach $1.6T by 2025. Explore key growth drivers, therapy areas, and industry challenges.

An in-depth study of pharmacy management systems updated for 2026. Explore key software features, compare top vendors (RedSail, Outcomes/TDS, Epic, Oracle Health), and learn about AI, DSCSA compliance, and market consolidation trends.

Learn about Patient-Reported Outcomes (PRO) systems in clinical trials. This guide covers ePRO data collection, PROMs, FDA PFDD guidelines, CONSORT 2025 updates, AI-driven ePRO platforms, and challenges

Updated 2026 guide to AI-assisted surgery companies including Intuitive Surgical's da Vinci 5, Medtronic Hugo (FDA-cleared Dec 2025), CMR Versius Plus, Stryker Mako 4, and more. Market data, clinical outcomes, and regulatory trends.

An in-depth analysis of the ICH E6(R3) Good Clinical Practice guidelines for 2025. Explore key changes in Quality by Design, data governance, and decentralized

An in-depth guide to ICH Q10 Pharmaceutical Quality Systems (PQS), updated for 2026 with ICH Q9(R1), Q12-Q14 developments, FDA QMM program updates, and AI-driven quality management trends.

Updated 2026 guide to synthetic data in pharmaceutical research. Covers acceptance criteria for fidelity, utility, and privacy, plus latest FDA/EMA AI guidance, EHDS, and diffusion model advances.

An overview of value-based contracting (VBC) in pharmaceuticals, where drug prices are tied to patient outcomes. Covers models, challenges, case studies, CMS CGT Access Model (2025), finalized VBP rule (2026), and IRA drug price negotiations.

Updated 2026 guide for IT on 21 CFR Part 11. Covers FDA CSA guidance, AI compliance, electronic records & signatures, system validation, audit trails & data integrity

Learn best practices for writing an effective FDA 483 response. Updated with FY2024-2025 enforcement data and FDA's AI-driven inspection targeting. Covers CAPAs, timelines, and strategies to avoid Warning Letters.

An analysis of the 2025 surge in new drug manufacturing plants from Eli Lilly, AstraZeneca & more. Learn about the key drivers: supply chain & geopolitics.

A comprehensive guide to pharmaceutical licensing deals updated for 2026. Covers deal structures, co-development agreements, financial terms, milestone payments, royalty rates, and the record $250B+ licensing market with China-sourced innovation trends.

A detailed profile of Cognition Therapeutics (CGTX) analyzing its pipeline, financials, and lead drug zervimesine (CT1812), a sigma-2 modulator for Alzheimer's and DLB, with 2025-2026 updates including Phase 3 plans, SHIMMER publication, and START trial enrollment.

Updated 2026 guide to HCP marketing in the pharmaceutical industry. Covers AI-driven personalization, omnichannel engagement, digital strategies, mobile-first content, and the latest FDA and IFPMA regulatory developments.

Explore Salesforce Health Cloud and Agentforce Life Sciences for biopharma. Updated for 2026 with Life Sciences Cloud GA, Agentforce AI, and new customer wins from Novartis and AstraZeneca.

Learn the key updates in ISPE GAMP 5 Second Edition. Updated for 2026 with FDA's finalized CSA guidance, ISPE GAMP AI Guide, and EU Annex 11/22 revisions

An in-depth analysis of Merck's GPTeal platform and expanding AI strategy. Covers the KERMT drug discovery model, Mayo Clinic partnership, CSR automation results, and enterprise-scale LLM deployment across pharma R&D.

An in-depth case study of AstraZeneca's ChatGPT and generative AI implementation, updated for 2026. Covers enterprise strategy, R&D use cases, self-healing supply chains, and agentic AI adoption.

Learn the end-to-end drug development pipeline, from initial drug discovery and preclinical research to Phase I-IV clinical trials and final FDA approval. Updated for 2026 with latest success rates, AI drug discovery progress, and regulatory developments.

Explore top MS in AI for Drug Development programs for 2025. This guide reviews curricula, career prospects, and leading universities like UCSF and Maryland.

Updated 2026 analysis of top Learning Management Systems (LMS) for biotech and life sciences. Compare validated platforms for GxP, 21 CFR Part 11 & ICH E6(R3) compliance.

Learn how to run private LLM inference for biotech. Updated for 2026 with NVIDIA Blackwell B200, Llama 4, FDA AI guidance, EU AI Act compliance, and on-premise deployment for data privacy, security, HIPAA compliance, and cost analysis

Explore Bespoke Labs' specialized open-source LLMs including MiniChart (charts), MiniCheck (fact-checking), Stratos, and the OpenThinker family from v1 through OpenThinker3 and Agent models.

Learn about OpenAI's Stargate project, a $500B AI infrastructure initiative. Updated for 2026 with the latest on datacenter locations, custom chips, global expansion, and progress.

See the 2025 list of the most expensive drugs in the USA, led by multi-million dollar gene therapies like Lenmeldy & Elevidys for rare diseases. Learn why price

Learn how spec-driven development (SDD) and tools like GitHub Spec Kit, AWS Kiro, and Tessl improve AI code generation. Updated for 2026 with the latest SDD ecosystem developments.

A summary of OpenAI DevDay 2025 announcements. Learn about new models like GPT-5 Pro and Sora 2, developer SDKs, and ChatGPT's evolution into an AI platform.

An overview of Anthropic's Claude Sonnet 4.5 model, its performance benchmarks, Claude Code 2.0 features like checkpoints, subagents, hooks, and IDE integration, plus updates on Opus 4.6 agent teams

Updated 2026 guide to FDA animal drug regulation: how CVM approves veterinary drugs, key differences from human drugs, recent approvals, antimicrobial stewardship, and telemedicine rules.

A comprehensive 2026 index of open-source LIMS with details on each system's license, technology stack, latest releases, and intended use for clinical, research, and biobanking labs.

A guide to the top 10 specialty pharmacy management platforms in 2025-2026. We compare systems from WellSky & McKesson to AI-powered tools like House Rx, Mandolin & Lamar Health for prior authorization & patient care

A 2026-updated review of top AI chatbots in healthcare. Covers Ada Health, Infermedica, Wysa, ChatGPT, and lessons from Babylon's collapse and Woebot's shutdown. Includes FDA regulatory developments and market trends.

An overview of IBM's Granite 4.0 LLM family, including its hybrid Mamba-2/Transformer architecture, Nano edge models, Granite Vision, Granite Guardian safety tools, and applications for healthcare AI and data privacy.

Analysis of AI's role in hospital operations for 2025, covering automated documentation, workflow efficiency, and reduced physician burnout with new data and ca

Analysis of the $90B OpenAI-AMD strategic partnership for 6GW of MI450 GPUs, including stock warrants, MI450 specs (CDNA 5, 2nm, HBM4), and OpenAI's 33GW multi-vendor compute strategy

An overview of COMS Interactive's DaylightIQ Clinical Outcomes Management System for long-term care. Learn how evidence-based protocols, AI-powered tools, and PointClickCare integration improve patient outcomes across 30,000+ provider organizations.

Learn about AI company Cohere, its enterprise focus, Command family of LLMs, North agent platform, and path to a potential 2026 IPO. Updated with $240M ARR, Tiny Aya models, and latest developments.

A data-driven analysis of the US biotech job market in 2025. Learn about employment trends, the skills gap, top geographic hubs, and the industry's future outlo

Learn how to create apps for ChatGPT in 2025. This guide explains the OpenAI Apps SDK, the GPT Store, custom GPTs, and provides a step-by-step developer workflo

Updated for 2026, this article profiles five leading technology companies in healthcare AI—IntuitionLabs, AWS, Google Cloud, IBM, and Microsoft—examining their latest solutions for clinical workflows, AI agents, and patient engagement.

Learn best practices to evaluate CRO costs for your clinical trial budget. This 2026-updated guide covers RFPs, pricing models (fixed, T&M, FSP), benchmarking, and AI-driven cost optimization.

A detailed overview of IQVIA's technology solutions for life sciences, updated for 2026. Covers data management, OCE-to-Salesforce Life Sciences Cloud transition, AI agents with NVIDIA, analytics platforms, and RWE.

Learn how to build robust LLM evaluation frameworks for biotech. Updated for 2026 with GPT-5, Med-Gemini, HealthBench results, FDA guidance, and the latest biomedical benchmarks including BLUE, BLURB, and hallucination detection frameworks.

An overview of Next Best Action (NBA) in pharma marketing. Learn how AI-driven NBA strategies optimize HCP engagement, with 2026 updates on agentic AI, Salesforce Agentforce, and the PharmaForceIQ-Aktana acquisition.

An analysis of China's open-source LLM landscape in 2025. Covers key models like Qwen, Ernie, and GLM from major tech firms and leading AI startups.

Learn the hardware requirements for running OpenAI's GPT-OSS-20B model locally. Updated for 2026 with RTX 5090/5080 coverage, GDDR7 GPUs, and the latest inference frameworks.

An examination of the five key technical innovations behind ChatGPT, from the Transformer architecture and pretraining to RLHF and its successors (DPO, GRPO), GPU hardware evolution through NVIDIA Blackwell, and tokenization.

An analysis of leading software for molecular modeling and simulation. Learn about features, theoretical methods (MD, QM), performance, and use cases.

Learn about Apache Airflow's core architecture (including Airflow 3.x features like DAG versioning, Task Execution API, and HITL workflows), and its application for building data workflows in life sciences.

An in-depth technical comparison of five cheminformatics platforms (RDKit, ChemAxon, OpenEye, Schrödinger, BIOVIA), evaluating SAR/QSAR, ADMET prediction, virtual screening, and chemical library management. Updated for 2026 with Certara-ChemAxon acquisition, ROCS X launch, and AI integration trends.

Examines the 2025 AI regulatory frameworks for biopharma in the U.S., EU, U.K., and Canada. Details compliance obligations for GenAI, GxP, and SaMD.

Learn the architectural differences between event sourcing and queue-based systems. Covers immutable event logs, data traceability, replayability, and 2025–2026 tooling ecosystem maturity including Kurrent, Kafka KRaft, and cloud-native event services.
Comprehensive guide to Mixture of Experts (MoE) models, covering architecture, training, and real-world implementations including DeepSeek-V3, Llama 4, Mixtral, and other frontier MoE systems as of 2026.

An educational overview of hospice EMR systems, including essential features for documentation, scheduling, billing, HIPAA compliance, and the mandatory HOPE assessment tool transition effective October 2025.

Updated guide to GAMP 5 guidelines for validating computerized systems, covering the 2025 ISPE GAMP AI Guide, FDA CSA final guidance, EU Annex 11/22 drafts, and risk-based lifecycle approach.

Learn about the specialized software tools used across the drug development lifecycle, from discovery and preclinical research to manufacturing and commercialization. Updated for 2026 with ICH E6(R3), DSCSA compliance deadlines, IDMP/PMS timelines, and the latest in AI-driven drug design.

An updated analysis of leading Regulatory Information Management (RIM) systems for 2026, covering Veeva Vault RIM, IQVIA SmartSolve RIM, ArisGlobal LifeSphere, Ennov, MasterControl, EXTEDO, and others, with the latest on eCTD 4.0, IDMP compliance, and AI-powered automation.

Updated for 2026, this article provides a technical overview of pharmacovigilance software, detailing core functions, regulatory compliance, agentic AI developments, and a comparison of leading platforms including Oracle Argus, ArisGlobal LifeSphere, Veeva Vault Safety, and more.

Learn what Policy as Code (PaC) is and why it is used in healthcare to automate enforcement of security, privacy, and compliance regulations, including the latest HIPAA Security Rule updates and OPA 1.0 developments.

Examines how to apply Git version control workflows to satisfy FDA compliance under the 2026 QMSR, covering traceability, audit trails, CSA guidance, SBOM requirements, and standards like 21 CFR Part 11 & IEC 62304.

Learn about FHIR (Fast Healthcare Interoperability Resources), the modern HL7 standard for exchanging health data using web APIs and self-contained resources. Updated for 2026 with FHIR R6 ballot, TEFCA FHIR exchange, CMS-0057-F, and EU EHDS developments.

An overview of AI agents in pharmacovigilance (PV), covering agentic AI, generative AI, and ML applications in safety data processing, ICSR management, and signal detection, with updates on the CIOMS WG XIV final report, FDA's Elsa tool, and EU AI Act requirements.

A comprehensive overview of the CMS definition for TPMOs, their role in the Medicare enrollment chain, and evolving regulations from CY 2023 through the CY 2027 proposed rule, including the Texas court injunction on CY 2025 compensation reforms.

A comprehensive guide to the IEC 62304 standard for medical device software, updated for 2026. Covers life cycle processes, safety classification, Edition 2.0 draft changes, FDA QMSR alignment, IEC 81001-5-1 cybersecurity, and regulatory compliance across FDA and EU MDR.

An analysis of top ERP systems for the pharmaceutical sector, evaluated on regulatory compliance (21 CFR Part 11), batch traceability, and serialization. Updated for 2025–2026 with DSCSA enforcement timelines, AI/Copilot capabilities, and latest platform releases.

Examine the role of AI in Health Information Exchange (HIE), including TEFCA growth, FHIR mandates, data standardization, security, predictive analytics, and ethical considerations as of 2026.

This article explains the core functions of LIMS for sample management, data integrity, and regulatory compliance, and provides a comparative analysis of top systems.

Learn methods for integrating ChatGPT with private enterprise data using Microsoft Azure and Microsoft Foundry. Covers security controls, compliance, Azure OpenAI Service, agents, MCP, and agentic retrieval.

Learn about the FDA Adverse Event Reporting System (FAERS), now updated in real time with over 31 million reports, for post-marketing drug safety surveillance, signal detection, and AI-enhanced pharmacovigilance.

Comprehensive guide to Software as a Medical Device (SaMD): IMDRF definition, FDA QMSR and PCCP frameworks, EU MDR/AI Act compliance, real-world examples, and 2025-2026 regulatory updates.

Learn about Clinical Decision Support (CDS) systems, from early rule-based expert systems to modern data-driven models powered by artificial intelligence. Updated for 2026 with the latest FDA clearances, EU AI Act implementation, and industry developments.

This article defines open-source LIMS and details its core functions, such as sample registration, data collection, quality control, and workflow management. Updated for 2026 with SENAITE 2.6.0, LabKey 25.11, OpenELIS AI features, FDA CSA guidance, and current market data.

Comprehensive review of global Health Management Information Systems (HMIS), covering major platforms (Epic, Oracle Health, DHIS2, OpenMRS, OpenEMR), AI integration, cloud adoption, interoperability standards, and 2025-2026 developments.

An overview of the healthcare revenue cycle, a financial process covering all administrative and clinical functions from patient scheduling to payment collection.

An explanation of Clinical Data Management (CDM) and its function in research. Learn how CDM ensures high-quality, reliable data for clinical trials.

Profiles of leading US researchers and industry pioneers applying generative AI to pharmaceutical R&D, drug discovery, protein design, and clinical trials – updated with 2025-2026 milestones including Phase II/III results, major funding rounds, and regulatory breakthroughs.

Learn about AHIP certification, the standard for Medicare agents. Updated for 2026 with current costs, NABIP alternative, IRA impacts, and Medicare Advantage market trends.

An analysis of AI adoption trends in U.S. hospitals through 2025-2026. Examines clinical and operational use cases including ambient AI scribes, Epic's new AI tools, FDA regulatory updates, and ethical considerations in healthcare.

Examines the top 10 programming languages by usage and popularity in the context of AI-assisted coding, analyzing AI tool support (Copilot, Cursor, Claude Code, Amazon Q Developer) and ecosystem maturity with 2025-2026 data.

An analysis of the 2025 healthcare administration job market. Examines key trends, demand drivers, worker shortages, and BLS employment projections.

An analysis of large language model (LLM) diagnostic performance compared to physicians, covering GPT-4o, MedGemma, meta-analyses on LLM-assisted diagnosis, patient trust surveys, and 2025-2026 regulatory developments.

Learn about mechanistic interpretability, named an MIT 2026 Breakthrough Technology. Covers circuit tracing, sparse autoencoders, attribution graphs, and how researchers are reverse-engineering AI models to uncover causal mechanisms within neural networks.

A comprehensive guide to pharmaceutical KPIs for quality, compliance, and operational excellence. Covers metrics for R&D, clinical trials, manufacturing, supply chain, and pharmacovigilance, updated with 2025-2026 regulatory developments including FDA QMM, ICH E6(R3), and ISO 9001:2026.

An analysis of the core technologies and design philosophies of Wolfram Alpha (symbolic AI) and ChatGPT (generative AI), detailing their key differences. Updated for GPT-5.2, Wolfram Language 14.3, and the transition from plugins to Custom GPTs.

This article explains how pill identifier software improves patient safety by accurately identifying medication using physical traits, AI-powered image recognition, and deep learning models achieving over 96% accuracy, preventing costly medical errors.

An educational profile of ZUSDURI, a mitomycin hydrogel for intravesical chemoablation of low-grade, intermediate-risk non-muscle invasive bladder cancer (NMIBC). Updated with 2026 J-code reimbursement, UGN-103 UTOPIA trial results, and commercial launch data.

An analysis of the Medicare program in 2025, detailing the structure and the significant policy impacts of the One Big Beautiful Bill Act (P.L. 119-21).

Examine 2025 trends for Contract Sales Organizations in pharma. This analysis compares top providers and strategies for HCP engagement and outsourcing.

This article explains how AI tools can validate regulatory dossiers, catch technical errors before submission, and minimize rework for emerging biotech companies. Updated with 2025-2026 developments including FDA's ELSA AI assistant, eCTD v4.0 validation updates, McKinsey benchmarks, and the latest Gemini 2.5 vs GPT-5 comparisons.

Examines the evolution of eCTD systems from specialized publishers for regulatory submissions to enterprise platforms that serve as a unified drug lifecycle archive, including the impact of eCTD v4.0, AI-powered RIM tools, and IDMP compliance.

Updated for 2026, this article examines the unique financial challenges facing regional hospitals — including 46% operating at negative margins — and details the accounting practices, software solutions, and AI-driven automation essential for maintaining stability and compliance.

An overview of San Francisco Bay Area neuroscience firms, updated February 2026. Examines companies in neurotechnology, BCIs, and neuropharmacology, with latest clinical trial results, FDA developments, and funding milestones.

A comprehensive overview of AI in pharmacovigilance (updated Feb 2026), covering agentic AI, GenAI-driven case processing, signal detection, CIOMS WG XIV framework, FDA/EMA joint principles, EU AI Act implications, and the latest industry platforms.

An overview of market access analytics covering its definition, scope, and role in helping pharmaceutical firms navigate complex payer reimbursement models. Updated for 2026 with IRA drug price negotiation impacts, agentic AI platforms, EU HTA harmonisation, and digital therapeutics reimbursement advances.

Learn to design, secure, and manage a HIPAA-compliant API. Updated for 2026 with the proposed Security Rule overhaul, mandatory encryption and MFA requirements, and current enforcement trends.

Learn how WORM (Write Once, Read Many) storage ensures data immutability and integrity for electronic records in the highly regulated biotechnology industry. Updated for 2026 with the latest on EU GMP Annex 11 revisions, SEC 17a-4 audit-trail alternatives, cloud WORM advances, and ransomware defense strategies.

Examine the GPT-5 model family's technical capabilities, ChatGPT Health and ChatGPT for Healthcare launches, applications in life sciences and medicine, and the ethical and regulatory considerations for AI in healthcare. Updated February 2026.

Examines the key scientific journals and trade publications in the pharma and biotech industries, covering peer-review, access models, and target audience.

An educational guide to executing compliant pharmaceutical remarketing. Explores platform policies on Google, Meta, LinkedIn, and Reddit, plus data privacy. Updated for 2025-2026 with Meta's health-data restrictions, FDA enforcement escalation, Google policy changes, and HIPAA tracking updates.

An overview of AI applications in the pharmaceutical sector, from generative AI to ML. Explains key IT management challenges like data, compliance, and security.

Updated 2026 survey of global online degrees and certificates in AI for pharmaceutical science. Compare programs by level, curriculum, cost, and duration – including new offerings from UCSF, Yale, and LIU.

An overview of OpenAI's open-weight GPT-OSS models. Examine their technical specifications, benchmark performance, gpt-oss-safeguard safety models, and applications for reasoning in healthcare.

Examines how AI accelerates the pharmaceutical drug pipeline, reducing time to market. Updated for 2026 with the latest clinical milestones, FDA guidance, and industry developments including Insilico Medicine's Phase IIa results and the Recursion-Exscientia merger.

Examines RegTech adoption challenges for emerging biotechs, focusing on cost, scalability, and AI-driven compliance. Covers flexible licensing, usage-based pricing, deferred-value models, and the latest platform innovations from Veeva Vault Basics and beyond.

An explanation of active learning principles and their adaptation for Large Language Models (LLMs) using human-in-the-loop (HITL) feedback for model alignment, including DPO, GRPO, and RLVR.

An overview of the pharmaceutical industry's history, from 19th-century origins to the current global market size, sales figures, and regional distribution.

An explanation of regulatory requirements for audit trails under 21 CFR Part 11 and EU Annex 11, covering automation strategies, data integrity measures, and the latest 2025-2026 regulatory developments including the Annex 11 draft revision and FDA CSA guidance.

A comprehensive guide to MCP servers for Claude Code web search, including Brave Search, Perplexity Sonar, Bright Data, and Firecrawl. Updated for 2026 with MCP Tool Search, OAuth authentication, and managed configuration.

An explanation of a single source of truth (SSOT) for the pharmaceutical industry. Learn how an SSOT integrates data across the drug lifecycle to ensure consistency.

An analysis of how CRM systems support medical device firms in managing HCP relationships, sales pipelines, and maintaining strict regulatory compliance.

An analysis of AI code assistants in large codebases. Evaluates GitHub Copilot, Amazon Q Developer, Cursor, Claude Code, Tabnine, and others on accuracy, context handling, security, and IDE integration. Updated February 2026.

This report details how pharmaceutical companies use mobile apps for patient support, medication adherence, and data collection, including the 2023-2024 digital therapeutics shakeout, FDA regulatory updates through 2026, and emerging AI-powered engagement strategies.

An overview of Reinforcement Learning (RL) and RLHF. Learn how RL uses reward functions and how RLHF incorporates human judgments to train AI agents. Updated with 2025-2026 developments including DPO, GRPO, DeepSeek-R1, and GPT-5.

Explore a comprehensive comparison of Tableau and Power BI, analyzing market share, features, pricing, and adoption trends for data professionals. Updated with 2025-2026 data including Gartner MQ 2025, Forrester Wave Q2 2025, and April 2025 pricing changes.

A technical review of dedicated OCR engines not based on LLMs. Examines computer vision and sequence modeling architectures, performance, and applications.

Explore how ERP systems integrate biotech R&D, manufacturing, and finance, ensuring GxP compliance and traceability. Covers major and niche vendor solutions.

A technical guide to Reinforcement Learning from Human Feedback (RLHF). This article covers its core concepts, training pipeline, key alignment algorithms, and 2025-2026 developments including DPO, GRPO, and RLAIF.

This article details AI applications in pharmaceutical business intelligence, covering drug discovery, clinical trials, supply chain, real-world evidence, and market intelligence.

Examines Kimi K2, a trillion-parameter open-weight LLM from Moonshot AI. Learn its technical details, development background, and strategic context.

This article explains Integrated Business Planning (IBP) as a strategic imperative for the pharmaceutical industry, detailing its role in unifying cross-functional plans and decision-making.

Explore 21 CFR Part 11 compliance for electronic records, signatures, and AI in GxP. Covers key elements, FDA guidance (including finalized CSA and AI credibility framework), and controls for data integrity and audit-ready systems. Updated February 2026.

This article defines Sales Force Effectiveness (SFE) in the MedTech industry, explaining its strategic importance for optimizing performance and achieving growth in a competitive market.

Explore key software needs, technology stacks, and specific tools like AI-driven drug design and cheminformatics in pharmaceutical software development.

Explore US pharmaceutical automation compliance, covering FDA regulations like cGMP & 21 CFR Part 11, Pharma 4.0 trends, challenges, and best practices.

Examines how an OpenAI AI system achieved a gold medal score at the 2025 IMO, detailing its performance, natural-language proofs, and AI reasoning ability. Updated with DeepMind's gold medal achievement and latest 2026 developments.

This article explains pharmaceutical serialization software, its critical role in securing the drug supply chain, and adherence to regulations like DSCSA and FMD. Updated with 2025-2026 enforcement deadlines and vendor developments.

Evaluate top 10 accounting/ERP solutions for pharmaceutical companies. Learn about features like regulatory compliance, batch tracking, and quality control systems.

Explore AWS cloud computing's role in life sciences for scalable data processing, HPC, and analytics. Learn how AWS facilitates innovation in biotech and pharma.

Learn how Power BI consultants apply business intelligence to pharmaceutical data, addressing regulatory needs, clinical trial insights, and RWE analysis.

Explore AI code assistants suitable for air-gapped, on-premises enterprise deployment. Understand infrastructure, security, and integration for highly regulated environments.

This article details AI's role in biotech sample management, covering traditional workflows, challenges, AI innovations, regulatory issues, and future outlook.

Explore clinical AI's role in patient care, decision-making, and medical data analysis. Learn about its applications in diagnosis, treatment, and outcome prediction, driven by tech advances.

Explore how Oracle Cloud CX supports customer experience in life sciences, encompassing CRM, marketing, and service applications for compliant engagement with HCPs and patients.

Explore Medidata Rave CTMS and EDC solutions, including their history, features, and real-world application in clinical trials. Learn about market standing and competitors.

Learn about 10 key AI innovations that optimize clinical trials, improving efficiency, reducing costs, enhancing patient safety, and speeding drug development.

Learn about key technical, regulatory, organizational, ethical, and financial barriers hindering AI adoption in life sciences, with emerging solutions including the latest FDA/EMA guidance and regulatory sandboxes.

Explore how generative AI is applied in mRNA vaccine development, using Moderna and Pfizer's COVID-19 vaccine as a case study to understand rapid immunization advancements.

Learn why new drug development takes over a decade, discussing the high attrition rates, extensive research, and regulatory hurdles involved in bringing medicines to market.

Learn how regulatory affairs ensures product compliance in health industries. Explore the fundamental role of AI and LLMs in modern regulatory processes, including the latest FDA/EMA joint guidance and EU AI Act requirements.

Learn about the 2025 job market for Computer System Validation (CSV) professionals. Understand CSV's critical role in regulated industries for data integrity and patient safety.

Learn about regulatory compliance software in pharmaceuticals, covering GMP, GLP, GCP, FDA 21 CFR Part 11, and EU Annex 11 standards for data integrity.

Learn about Pharmaceutical Field Force Effectiveness (FFE), its definition, strategic importance for commercial goals, and key performance indicators.

Explore pharmaceutical commercial operations, covering essential activities from market research and marketing strategy to sales execution and product distribution after regulatory approval.

Explore ChatGPT as a Generative AI and Large Language Model. Learn its core GPT architecture, Transformer backbone, and how it processes language.

This guide details a structured approach for Generative AI adoption in life sciences, covering strategy, governance, technology, training, and ethical considerations.

Learn how NetSuite ERP modules support U.S. pharmaceutical manufacturing labs, covering GMP, FDA (21 CFR Part 11) compliance, and integrations for quality and production. Updated for 2026 with NetSuite Next AI features and DSCSA compliance deadlines.

Compare GAMP 4 and GAMP 5. Learn key differences in their approaches to system validation, risk management, system classification, and integration of modern technologies. Updated with 2025 FDA CSA guidance and ISPE GAMP AI Guide.

Evaluate LMS platforms for life sciences. Understand key compliance requirements: FDA 21 CFR Part 11, GxP, electronic records, and audit trails for validated training.

Explore the distinct roles and benefits of Learning Management Systems (LMS), Digital Adoption Platforms (DAP), and Knowledge Management Systems (KMS) in life sciences.

Compares NetSuite and SAP ERP systems' technical capabilities and regulatory compliance features for pharmaceutical and life sciences companies.

Learn about leading pharmaceutical market intelligence firms, their data analysis methods, and services like drug pipeline tracking, sales forecasts, and regulatory insights.

This article lists 10 free generative AI courses for pharmaceutical professionals. Learn LLMs, prompt engineering, and AI applications in drug R&D.

Learn how ICD-10 codes, essential for healthcare data in EHRs, are transformed into numerical embedding vector spaces for machine learning and data science applications.

Explore the history and development of Anthropic's Claude 4 large language model, covering its evolution to Claude 4.5, key features, benchmarks, and advancements through January 2026.

This article compares the user interfaces of leading conversational AI tools like ChatGPT, Gemini, and Claude, detailing their design, features, and professional impact for business users.

Learn about 21 CFR Part 11, the FDA regulation for electronic records and signatures. Updated for 2026 with the latest FDA guidance on electronic systems, Computer Software Assurance (CSA), and GAMP 5 Second Edition.

Explore Good Manufacturing Practice (GMP) in pharma. Learn its principles, global regulations, implementation, and compliance challenges for quality assurance.

This guide explains GAMP 5, its principles, and lifecycle for computerized system validation in pharma, covering regulatory compliance and real-world applications. Updated for 2025-2026 with FDA CSA guidance, EU Annex 11 revision, and ISPE AI Guide.

Learn what Computer System Validation (CSV) is, its crucial role in pharmaceutical and biotech compliance, ensuring data integrity and regulatory adherence for patient safety.

Compares leading AI OCR models and tools for PDF to structured text conversion in 2026. Examines open-source and commercial solutions including Mistral OCR 3, GPT-4o, and cloud services.

This report details deploying LLMs on 24GB GPUs, covering model architectures, VRAM needs, and optimization methods for efficient local operation.

Learn about meta-prompting, an advanced technique using LLMs to generate, modify, and optimize their own prompts, enabling iterative and complex task execution.

Explore profiles of top global pharmaceutical thought leaders, including their backgrounds, speaking topics, notable engagements, and industry influence.

Learn to build a pharmaceutical manufacturing plant from concept through commissioning. This guide covers planning, regulatory compliance, design, equipment, quality, validation, and approval.

Explore how AI-powered computer vision is transforming pharmaceutical quality control. Review top vendors, applications, and trends in pharma QC automation.

How AI leaders like Amodei, Hassabis, and Huang are accelerating drug discovery, genomics, and life science breakthroughs—compressing decades into years. Updated January 2026 with AlphaFold 3, Claude for Life Sciences, NVIDIA Evo 2, and $3B+ pharma partnerships.

Comprehensive guide to BI and dashboard tools for pharma: Power BI, Tableau, Qlik, Looker, Domo, Sisense, Strategy (MicroStrategy), IBM Cognos. Updated pricing, compliance, and adoption trends for 2025-2026.

How Veeva Crossix and big data are transforming pharma marketing analytics: capabilities, compliance, case studies, and adoption trends in the U.S.

Guide to MarTech API integrations for pharma: CRM, marketing automation, analytics, consent, compliance, and best practices for unified digital engagement.

In-depth guide to U.S. pharmaceutical marketing regulations: FDA, FTC, Sunshine Act, anti-kickback, compliance, and IT obligations for pharma companies.

A practical guide to clinical trial phases 1–3 for pharma IT: objectives, compliance, costs, and IT infrastructure for modern drug development.

Guide to outsourcing HCP marketing in pharma: benefits, risks, industry trends, compliance, and how to manage third-party partnerships effectively.

Best practices for pharma teams to secure first meetings with HCPs: outreach tactics, channel comparison, overcoming gatekeepers, and expert tips for success.

Overview of major HCP data providers, U.S. compliance rules, and best practices for pharma IT. Includes vendor comparison, legal requirements, and governance tips.

How biotech and pharma companies use NetSuite for compliance: FDA 21 CFR Part 11, GxP, HIPAA, SOX, audit trails, e-signatures, and reporting best practices.

Evidence-based UX strategies for digital HCP engagement platforms in pharma: trends, challenges, best practices, and compliance for IT teams.

Comprehensive comparison of Power BI and Tableau for pharma: features, pricing, compliance, and use cases for IT and analytics teams.

A technical comparison of AI agents and AI workflows in pharmaceutical IT, with use cases, pros and cons, and adoption trends for U.S. pharma.

A technical overview of remote detailing in the pharmaceutical industry: adoption trends, technology platforms, compliance, and best practices.

A detailed survey of large language model benchmarks in life sciences, covering biomedical NLP, drug discovery, and genomics, with industry use cases and top model performance.

A comprehensive review of software solutions for optimizing pharmaceutical field sales routes, comparing CRM, SFA, and mapping tools for efficiency, compliance, and analytics.

A detailed comparison of Windsurf (Codeium), Cursor, and GitHub Copilot for enterprise software development in the pharmaceutical industry, focusing on security, compliance, and productivity. Updated January 2026 with latest pricing, features, and the Cognition acquisition of Windsurf.

An exploration of how artificial intelligence is revolutionizing drug development processes, from target identification to clinical trials, with focus on implementation strategies and success metrics.

An analysis of how artificial intelligence is transforming regulatory affairs in pharmaceuticals, from submission preparation to compliance monitoring and regulatory intelligence.

A technical guide to developing customized CRM solutions for pharmaceutical companies using AI-assisted development tools, focusing on compliance, data security, and industry-specific requirements.

Detailed case studies examining successful implementations of Randomization and Trial Supply Management systems in U.S. clinical trials, highlighting best practices and measurable outcomes.

A comprehensive analysis of ChatGPT integration in life sciences, examining implementation strategies, regulatory compliance, and real-world applications across pharmaceutical research and development.

An in-depth analysis of the biotech ecosystem in the San Francisco Bay Area, examining key players, emerging startups, investment trends, and innovation clusters as of January 2026. Updated with latest data on 23andMe bankruptcy, major acquisitions, and 2025 layoffs.

Exploring how modern RTSM solutions are evolving to improve patient experience in clinical trials, featuring innovative approaches to recruitment, engagement, and trial management.

A detailed comparison of key compliance frameworks in pharmaceutical IT, including FDA 21 CFR Part 11, GDPR, HIPAA, and GxP, with implementation strategies and best practices.
An evaluation of RAG systems' effectiveness in processing pharmaceutical documentation, analyzing accuracy, compliance adherence, and practical applications in drug development and clinical trials.

An in-depth comparison of cloud-based and on-premise Randomization and Trial Supply Management (RTSM) solutions, analyzing security, compliance, cost, and operational considerations for pharmaceutical companies.

A comprehensive analysis of the remote patient monitoring landscape in the US healthcare system, examining technological advances, regulatory framework, and implementation challenges in 2025.

A curated guide to the most reliable and comprehensive pharmaceutical industry news sources, specifically tailored for IT professionals working in pharma and life sciences.

An in-depth analysis of how artificial intelligence is transforming clinical data management across US healthcare, from EHR documentation to clinical trials and real-world evidence.

A comprehensive guide to integrating Randomization and Trial Supply Management (RTSM) systems with Electronic Data Capture (EDC) platforms, covering benefits, challenges, and vendor solutions.

An in-depth analysis of top Clinical Research Management Systems in the United States, comparing features, benefits, and implementation strategies for pharmaceutical companies.

Explore how MCP is revolutionizing data integration and AI applications in pharmaceutical research, clinical trials, and healthcare systems for enhanced compliance.

A comprehensive guide to regulatory requirements and best practices for implementing compliant Randomization and Trial Supply Management (RTSM) systems in clinical trials, covering FDA, EMA, and global standards.

A comprehensive guide to how Amazon Web Services (AWS) is transforming pharmaceutical operations from drug discovery to manufacturing, with real-world case studies from Pfizer, Moderna, Merck, and more.

Explore real-world case studies of how pharmaceutical companies are leveraging big data, AI, and cloud computing across the drug lifecycle - from discovery to marketing - with measurable outcomes and lessons learned.

A comprehensive guide to electronic patient record systems in pharmaceutical research and clinical trials, exploring their benefits, implementation challenges, and regulatory considerations.

A comprehensive exploration of generative AI proof of concepts in pharmaceutical research, examining real-world applications, implementation strategies, and measurable outcomes across the drug development pipeline.

A comprehensive analysis of how Google Cloud Platform (GCP) is revolutionizing pharmaceutical operations, from AI-powered drug discovery to clinical trial management and regulatory compliance.

An in-depth analysis of IBM's contributions to pharmaceutical innovation, from AI-powered drug discovery and hybrid cloud infrastructure to regulatory compliance and security solutions.

A comprehensive analysis of the life sciences job market in 2025, exploring emerging roles, skill requirements, salary trends, and career opportunities across pharmaceutical, biotechnology, and medical device sectors.

An in-depth exploration of how pharmaceutical companies leverage Microsoft Azure's cloud platform for drug discovery, clinical trials, manufacturing, and regulatory compliance, with real-world case studies and implementation strategies.

An in-depth guide to designing and implementing modern datacenter infrastructure for pharmaceutical companies, focusing on scalability, security, regulatory compliance, and integration with cloud services for drug development and manufacturing.

A comprehensive guide to implementing and optimizing NetSuite ERP for pharmaceutical companies, covering supply chain management, manufacturing operations, quality control, regulatory compliance, and integration with specialized pharma systems.

A comprehensive analysis of how pharmaceutical and biotech companies are leveraging NVIDIA's latest H100 and Blackwell GPUs to accelerate drug discovery, protein structure prediction, and AI-driven research, with detailed case studies from leading companies.

Comprehensive guide on RTSM best practices for Phase 3 trials, covering randomization strategies, global supply chain management, regulatory considerations (FDA, ICH E6(R3)), system integration, risk mitigation, and future trends including AI-driven forecasting and decentralized trial support.

Comprehensive analysis of big data technologies used in pharmaceutical industry, including Hadoop, Spark 4.x, cloud data warehouses (Snowflake, Databricks), NoSQL databases, and specialized genomics platforms, with detailed comparisons and implementation examples. Updated for 2025-2026 with latest market data and technology developments.

Comprehensive guide on implementing CAPA dashboards in pharmaceutical quality management, covering data requirements, key metrics, dashboard design principles, and implementation in Power BI, Tableau, and Google Data Studio.

Comprehensive overview of how computer vision technologies are revolutionizing pharmaceutical quality control processes, from tablet inspection to packaging verification, with real-world implementation examples and ROI analysis.

Comprehensive overview of Oracle's role in the pharmaceutical industry, covering their Health Sciences solutions, cloud infrastructure, compliance features, and case studies of successful implementations at major pharma companies.

Practical guide for Market Access teams and field representatives on handling common payer objections in oncology, including strategies for verbal and written communications, real-world examples, and response frameworks.

A comprehensive analysis of how pharmaceutical companies leverage SAP's enterprise solutions for drug development, clinical trials, manufacturing, supply chain management, and regulatory compliance, with detailed case studies from leading pharma companies.

A comprehensive analysis of leading medical technology companies worldwide that are at the forefront of AI adoption, examining their innovative applications in medical imaging, diagnostics, robotic surgery, patient monitoring, and personalized medicine, with detailed profiles of each company's AI technologies and market impact.

A comprehensive guide to building effective performance dashboards for small hospitals, covering key metrics, data integration, visualization tools, and practical implementation strategies to improve decision-making and operational efficiency.

An in-depth analysis of the leading AI consulting firms serving U.S. pharmaceutical companies, highlighting their focus areas, technologies, and recent activities in drug discovery, compliance, efficiency, and commercial applications.

A comprehensive guide for pharmaceutical marketing teams to plan, build, and optimize a custom HCP engagement portal to boost physician engagement through educational content, interactive tools, and personalized experiences. Updated for 2026 with AI, Vault CRM, and regulatory insights.

A comprehensive guide to Key Opinion Leader (KOL) tiering in pharmaceutical marketing, covering definition, strategic importance, tiering criteria, engagement strategies, AI-powered platforms, and compliance considerations for U.S. pharma companies. Updated for 2026.

A detailed exploration of Non-Personal Promotion strategies in pharmaceutical marketing, covering best practices, compliance requirements, and tools for effective HCP engagement without face-to-face interaction.

A comprehensive step-by-step guide for pharmaceutical marketing teams to plan, develop, and launch effective patient portals that improve engagement, adherence, and outcomes while ensuring regulatory compliance.

A comprehensive analysis of Workday's growing adoption in the pharmaceutical and life sciences sector, examining market trends, key drivers, notable industry users, competitive positioning against SAP, Oracle, and ADP, and future outlook for enterprise cloud solutions in life sciences.

An in-depth exploration of how data science is revolutionizing the life sciences industry, from drug discovery to clinical trials, with real-world applications and case studies. Updated January 2026 with latest FDA AI guidance, Insilico Medicine Phase IIa results, and major industry consolidations.

Learn how to identify, segment, and engage Healthcare Professionals (HCPs) and Key Opinion Leaders (KOLs) effectively for pharmaceutical commercialization, including methodologies, data sources, tools, and compliance considerations.

A comprehensive guide to compliant pharmaceutical sales practices, covering FDA regulations, PhRMA Code requirements, and best practices for engaging healthcare professionals while avoiding common compliance pitfalls.

A comprehensive guide to how pharmaceutical companies identify, segment, and engage healthcare professionals (HCPs) for sales outreach, covering databases, targeting strategies, digital tools, and compliance considerations.

A comprehensive overview of the most influential open-source software tools transforming pharmaceutical research, development, and manufacturing, from cheminformatics to clinical data management and regulatory compliance.

A comprehensive guide to the leading commercial analytics software platforms for pharmaceutical companies, covering sales forecasting, field force effectiveness, market access analysis, real-world evidence integration, customer segmentation, and omnichannel marketing optimization.

A comprehensive training manual for pharmaceutical sales representatives on using Veeva Vault CRM, covering account management, call logging, CLM presentations, sample distribution, Veeva AI Agents, and data synchronization on iPad, Android, and desktop. Updated for the 2025-2026 Vault platform transition.

A comprehensive analysis of cloud computing adoption versus traditional on-premises infrastructure in the pharmaceutical industry, including current adoption rates, historical trends, future forecasts, and key drivers of cloud migration.

A comprehensive guide to modern data warehousing solutions for life sciences organizations, covering cloud vs. on-premise strategies, technology stacks (Snowflake, Databricks, Redshift, BigQuery, Microsoft Fabric), compliance requirements, and scalable approaches for organizations of all sizes. Updated for 2026 with the latest platform features and market trends.

A comprehensive overview of Customer Relationship Management (CRM) platforms tailored for biotech companies, comparing various solutions and their features for compliance, sales, and customer relationship management in the life sciences sector.

A comprehensive guide to modern healthcare professional (HCP) engagement solutions for pharmaceutical companies, covering omnichannel strategies, compliance requirements, technology platforms, and best practices for tracking and measuring engagement effectiveness.

A comprehensive analysis of how pharmaceutical marketing differs from traditional industries, covering regulatory frameworks, key players, allowed practices, and compliance requirements in the U.S. healthcare market.

An in-depth analysis of the five most digitally innovative pharmaceutical companies in Europe, examining their AI initiatives, digital transformation strategies, and how they're leveraging technology to accelerate drug development and improve patient outcomes.

A comprehensive comparison of CRM requirements between pharmaceutical companies and other life sciences organizations, examining key differences in sales, marketing, compliance, and patient engagement approaches.

A comprehensive comparison of three approaches to adapting large language models for pharmaceutical applications: fine-tuning, distillation, and prompt engineering, with technical details and real-world examples.

A comprehensive analysis of Veeva's decision to end its partnership with Salesforce, exploring the strategic implications, timeline of events, and impact on life sciences companies facing this major industry transition.

In-depth analysis of Veeva Systems' transformation from 2021 to 2026, examining strategic shifts, product innovations including AI Agents, Vault CRM adoption, and continued market leadership in life sciences cloud solutions. Updated January 2026.

A detailed analysis of Veeva CTMS features and capabilities, exploring how it streamlines clinical trial operations and improves study execution.

A comprehensive technical guide for developers on creating custom dashboards in Veeva CRM using MyInsights, covering architecture, data access, and best practices.

A comprehensive guide to Veeva PromoMats features and capabilities, exploring how it streamlines promotional content management and ensures regulatory compliance in life sciences.

A comprehensive technical guide to Veeva Vault Platform, covering architecture, development capabilities, integration options, and security features for software developers.
© 2026 IntuitionLabs. All rights reserved.