
Analyze the May 2026 FDA drug repurposing initiative. Learn how AI indication discovery and real-world evidence shape regulatory pathways like 505(b)(2).

Analyze the May 2026 FDA drug repurposing initiative. Learn how AI indication discovery and real-world evidence shape regulatory pathways like 505(b)(2).
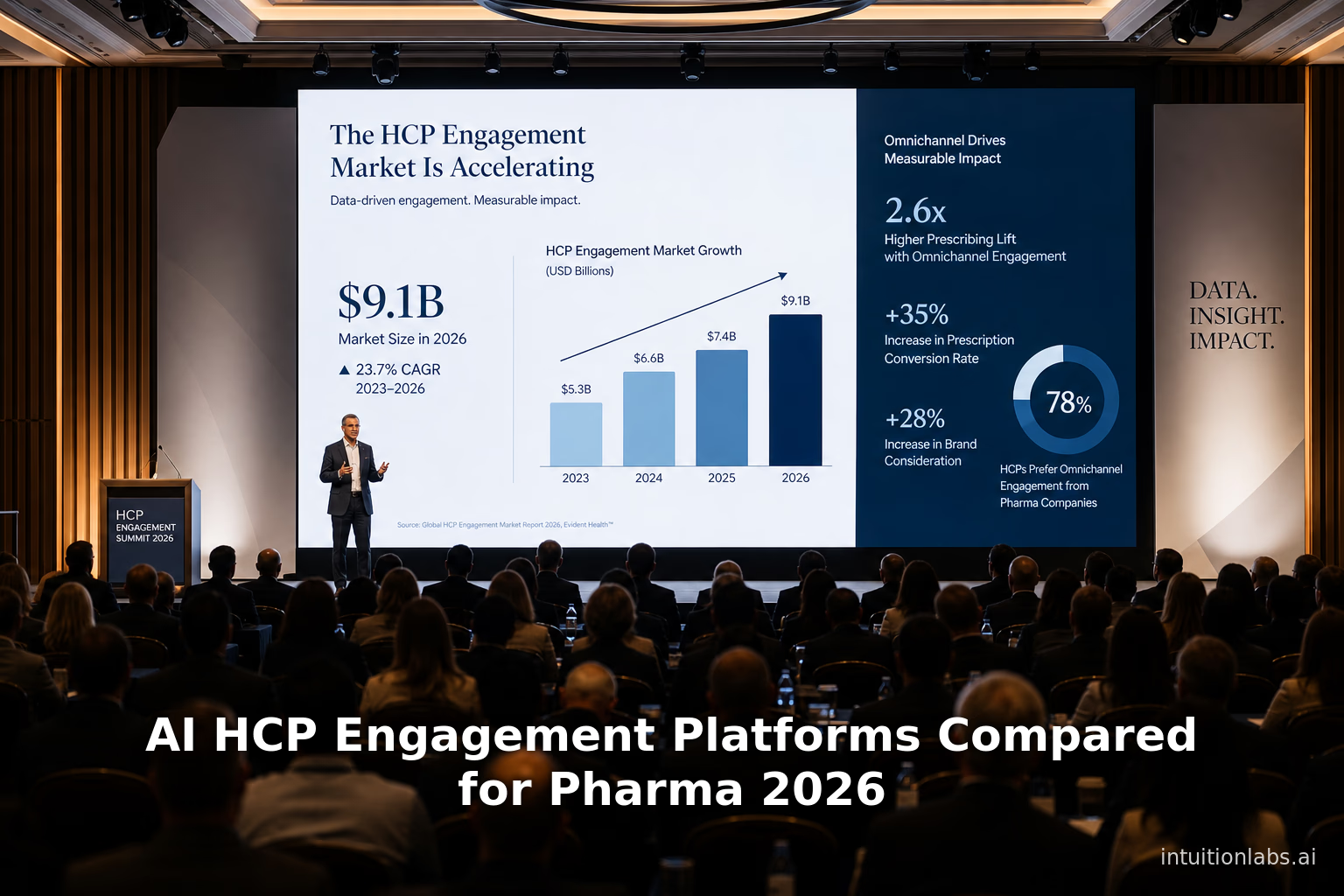
Compare leading AI HCP engagement platforms for pharma commercial operations. Review the 2026 capabilities of Aktana, OptimizeRx, Pega, and Veeva Vault CRM.

Explore how artificial intelligence is applied in pharmaceutical QC labs to automate OOS investigations, real-time batch release, and stability data analysis.

Examine the FDA's enforcement of ClinicalTrials.gov reporting mandates, historic compliance gaps among trial sponsors, and AI solutions for data disclosure.

Guide to designing an AI training program for pharmaceutical teams. Review role-based curriculum development, learning tools, and ROI measurement frameworks.

Analyze the FDA CDER 2026 guidance pipeline for artificial intelligence in drug manufacturing and digital health technologies in pharmaceutical development.

An overview of molecular pathology software, examining LIMS for NGS workflows, multi-omics data integration, and AI analysis tools in precision medicine.

Explore a 90-day diagnostic framework for assessing AI readiness in pharma. Learn how to evaluate data governance, regulatory compliance, and integration.

An in-depth analysis of the draft EU GMP Annex 22 guidelines for artificial intelligence in pharmaceutical manufacturing. Review AI compliance and risk rules.

Examine how smart manufacturing in pharma uses AI, digital twins, and IoT to optimize production, quality control, and supply chains using a GSK case study.

This article explains how artificial intelligence, advanced OCR, and LLMs extract unstructured clinical research and CRO data from complex PDF documents.

Examine the pharmaceutical AI strategy roadmap for scaling digital transformation. Learn about enterprise data infrastructure, LLMOps, and GxP compliance.

Examine how digital twins in clinical trials function as virtual control arms. This report reviews FDA guidance, AI models, and implementation requirements.

Analyze how FDA inspection delays and regulatory bottlenecks impact patient safety and drug supply. Learn how AI tools are addressing these critical backlogs.

Analyze the integration of AI with Lean Six Sigma in MedTech. Reviews DMAIC enhancements, regulatory compliance, and predictive quality management systems.

A technical guide to ChatGPT Enterprise deployment. Covers GPT-5 features, data privacy controls, security protocols, and employee training strategies.

Explore AI automation for Clinical Study Reports (CSRs). Analyze efficiency gains, regulatory compliance, and risks like hallucinations and data security.

Guide to AI-driven literature monitoring in pharmacovigilance. Covers NLP techniques, signal detection, and regulatory intelligence automation strategies.

Explore how AI automates CAPA and deviation workflows in Veeva Vault QMS. Covers NLP triage, ML root cause analysis, and regulatory compliance efficiency.

Analyze MLR review automation using Veeva PromoMats AI. Learn how Quick Check and Content Agents reduce compliance errors and streamline approval cycles.

Explore AI applications in clinical development plans, including protocol optimization, synthetic control arms, and patient recruitment strategies for trials.

Analyze the build vs buy AI decision in pharma. Compare costs, risks, and time-to-value for R&D and commercial teams to guide strategic investment.

Explore GxP audit trail requirements for AI systems. Review 21 CFR Part 11, Annex 11, and ALCOA+ rules for logging training data, prompts, and model outputs.

Learn how AI automates adverse event detection in pharmacovigilance. This guide covers GVP compliance, NLP methods, and validation standards for safety data.

Navigate FDA 21 CFR Part 11 for AI systems. This article details compliance strategies for validation, audit trails, and data integrity in regulated GxP setting

Learn how machine learning (ML) and AI are transforming pharmaceutical CMC process optimization in 2026. Covers applications, digital twins, regulatory updates (FDA/EMA 2026 AI principles, FRAME initiative), and real-world case studies.

Analyze the 2025 AI engineer job market. Learn the core responsibilities, average salary, and how the role differs from a data scientist or ML engineer.

Explore the AIME 2025 benchmark, a key test for AI mathematical reasoning. See how models like GPT-5 score over 94% and compare LLM performance on Olympiad-leve

An in-depth analysis of the HMMT25 AI benchmark for testing advanced mathematical reasoning in LLMs. See how models like Grok-4, GPT-5, and Gemini 3 perform on complex contest math problems, and how newer benchmarks like FrontierMath are raising the bar.

An overview of Next Best Action (NBA) in pharma marketing. Learn how AI-driven NBA strategies optimize HCP engagement, with 2026 updates on agentic AI, Salesforce Agentforce, and the PharmaForceIQ-Aktana acquisition.

An analysis of China's open-source LLM landscape in 2025. Covers key models like Qwen, Ernie, and GLM from major tech firms and leading AI startups.

An examination of the five key technical innovations behind ChatGPT, from the Transformer architecture and pretraining to RLHF and its successors (DPO, GRPO), GPU hardware evolution through NVIDIA Blackwell, and tokenization.

Updated guide to GAMP 5 guidelines for validating computerized systems, covering the 2025 ISPE GAMP AI Guide, FDA CSA final guidance, EU Annex 11/22 drafts, and risk-based lifecycle approach.

Examine the role of AI in Health Information Exchange (HIE), including TEFCA growth, FHIR mandates, data standardization, security, predictive analytics, and ethical considerations as of 2026.

Learn about Clinical Decision Support (CDS) systems, from early rule-based expert systems to modern data-driven models powered by artificial intelligence. Updated for 2026 with the latest FDA clearances, EU AI Act implementation, and industry developments.

An analysis of the core technologies and design philosophies of Wolfram Alpha (symbolic AI) and ChatGPT (generative AI), detailing their key differences. Updated for GPT-5.2, Wolfram Language 14.3, and the transition from plugins to Custom GPTs.

This article explains how AI tools can validate regulatory dossiers, catch technical errors before submission, and minimize rework for emerging biotech companies. Updated with 2025-2026 developments including FDA's ELSA AI assistant, eCTD v4.0 validation updates, McKinsey benchmarks, and the latest Gemini 2.5 vs GPT-5 comparisons.

A comprehensive overview of AI in pharmacovigilance (updated Feb 2026), covering agentic AI, GenAI-driven case processing, signal detection, CIOMS WG XIV framework, FDA/EMA joint principles, EU AI Act implications, and the latest industry platforms.

An overview of AI applications in the pharmaceutical sector, from generative AI to ML. Explains key IT management challenges like data, compliance, and security.

Updated 2026 survey of global online degrees and certificates in AI for pharmaceutical science. Compare programs by level, curriculum, cost, and duration – including new offerings from UCSF, Yale, and LIU.

Examines how AI accelerates the pharmaceutical drug pipeline, reducing time to market. Updated for 2026 with the latest clinical milestones, FDA guidance, and industry developments including Insilico Medicine's Phase IIa results and the Recursion-Exscientia merger.

This article details AI applications in pharmaceutical business intelligence, covering drug discovery, clinical trials, supply chain, real-world evidence, and market intelligence.

Explore 21 CFR Part 11 compliance for electronic records, signatures, and AI in GxP. Covers key elements, FDA guidance (including finalized CSA and AI credibility framework), and controls for data integrity and audit-ready systems. Updated February 2026.

Examines how an OpenAI AI system achieved a gold medal score at the 2025 IMO, detailing its performance, natural-language proofs, and AI reasoning ability. Updated with DeepMind's gold medal achievement and latest 2026 developments.

Learn about 10 key AI innovations that optimize clinical trials, improving efficiency, reducing costs, enhancing patient safety, and speeding drug development.

Learn how regulatory affairs ensures product compliance in health industries. Explore the fundamental role of AI and LLMs in modern regulatory processes, including the latest FDA/EMA joint guidance and EU AI Act requirements.

Explore ChatGPT as a Generative AI and Large Language Model. Learn its core GPT architecture, Transformer backbone, and how it processes language.

This article lists 10 free generative AI courses for pharmaceutical professionals. Learn LLMs, prompt engineering, and AI applications in drug R&D.

Learn about meta-prompting, an advanced technique using LLMs to generate, modify, and optimize their own prompts, enabling iterative and complex task execution.

An exploration of how artificial intelligence is revolutionizing drug development processes, from target identification to clinical trials, with focus on implementation strategies and success metrics.

An analysis of how artificial intelligence is transforming regulatory affairs in pharmaceuticals, from submission preparation to compliance monitoring and regulatory intelligence.

A technical guide to developing customized CRM solutions for pharmaceutical companies using AI-assisted development tools, focusing on compliance, data security, and industry-specific requirements.

A comprehensive analysis of ChatGPT integration in life sciences, examining implementation strategies, regulatory compliance, and real-world applications across pharmaceutical research and development.
An evaluation of RAG systems' effectiveness in processing pharmaceutical documentation, analyzing accuracy, compliance adherence, and practical applications in drug development and clinical trials.

An in-depth analysis of how artificial intelligence is transforming clinical data management across US healthcare, from EHR documentation to clinical trials and real-world evidence.

A comprehensive exploration of generative AI proof of concepts in pharmaceutical research, examining real-world applications, implementation strategies, and measurable outcomes across the drug development pipeline.

A comprehensive analysis of how Google Cloud Platform (GCP) is revolutionizing pharmaceutical operations, from AI-powered drug discovery to clinical trial management and regulatory compliance.

An in-depth analysis of IBM's contributions to pharmaceutical innovation, from AI-powered drug discovery and hybrid cloud infrastructure to regulatory compliance and security solutions.

A comprehensive analysis of leading medical technology companies worldwide that are at the forefront of AI adoption, examining their innovative applications in medical imaging, diagnostics, robotic surgery, patient monitoring, and personalized medicine, with detailed profiles of each company's AI technologies and market impact.

An in-depth analysis of the leading AI consulting firms serving U.S. pharmaceutical companies, highlighting their focus areas, technologies, and recent activities in drug discovery, compliance, efficiency, and commercial applications.

An in-depth exploration of how data science is revolutionizing the life sciences industry, from drug discovery to clinical trials, with real-world applications and case studies. Updated January 2026 with latest FDA AI guidance, Insilico Medicine Phase IIa results, and major industry consolidations.

A comprehensive guide to the leading commercial analytics software platforms for pharmaceutical companies, covering sales forecasting, field force effectiveness, market access analysis, real-world evidence integration, customer segmentation, and omnichannel marketing optimization.

An in-depth analysis of the five most digitally innovative pharmaceutical companies in Europe, examining their AI initiatives, digital transformation strategies, and how they're leveraging technology to accelerate drug development and improve patient outcomes.
© 2026 IntuitionLabs. All rights reserved.