
Learn what reinforcement learning (RL) is through clear explanations and examples. This guide covers core concepts like MDPs, agents, rewards, and key algorithm

Learn what reinforcement learning (RL) is through clear explanations and examples. This guide covers core concepts like MDPs, agents, rewards, and key algorithm

Learn what SharePoint Framework (SPFx) is, the client-side model for M365 development. Explore its history, architecture, and use with AI coding agents like Cla

A technical analysis of Google TPU architecture, from v1 to v7. Learn how this custom AI accelerator powers Gemini 3 with superior performance and efficiency vs

Learn why MSLs can discuss off-label data via scientific exchange while sales reps cannot, including FDA's 2023 SIUU guidance on firm-initiated scientific communications.

A comprehensive analysis of how data quality and data culture are foundational to AI success in pharmaceutical and life sciences organizations, covering assessment frameworks, governance models, regulatory compliance, and practical implementation roadmaps.

An analysis of private equity value creation for TPA & PBM companies post-acquisition. Learn key strategies for growth, operational efficiency, and M&A integrat
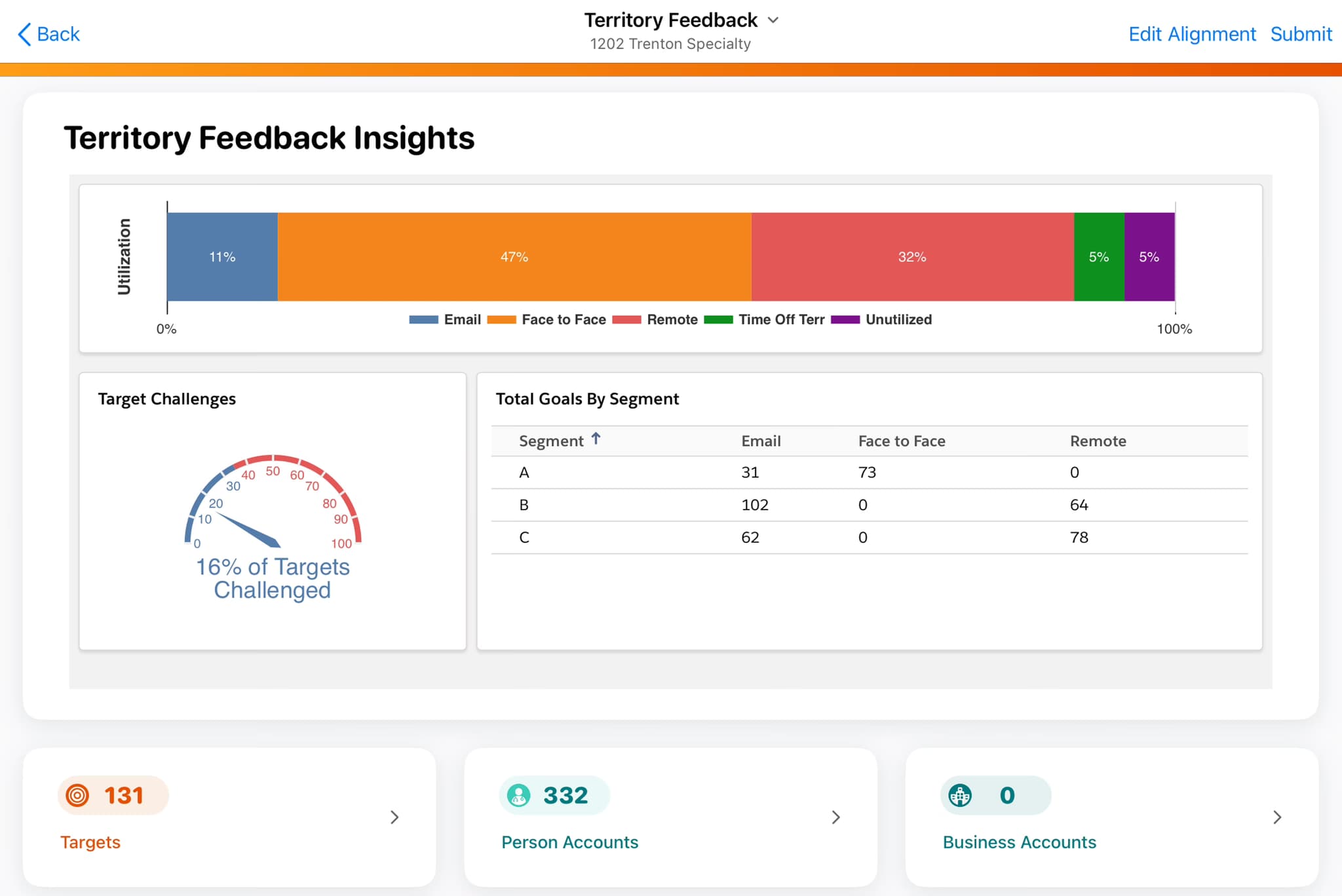
2026 guide to Veeva Vault CRM X-Pages development services. Covers X-Pages Studio vs custom code, Veeva AI Agent integration, Nitro data, timelines, costs, and how to select a certified development partner.

Learn the 5-stage eQMS maturity model for Biotech & MedTech startups. Updated April 2026 for the FDA QMSR (21 CFR 820 + ISO 13485:2016) effective Feb 2, 2026, CDER's third-year QMM pilot, and the latest Quality 4.0 AI trends.

This 2026 analysis reviews top patient payment platforms for automating collections. Compare features and performance of PatientPay, Cedar, RevSpring, and more.

Explore a detailed comparison of cash flow forecasting software for health systems. This 2026 analysis reviews StrataJazz, Obol, and Centage for hospital financ

A data-driven analysis of 2025 KLAS scores for top enterprise PACS vendors. Compare Sectra, Agfa, Fujifilm, and others on user satisfaction and performance.

Compare top healthcare CMMS platforms. This analysis reviews Accruent TMS, Brightly Biomed, and TRIMEDX RSQ on features, compliance, ROI, and user experience.

Learn the SCDC framework for differentiated messaging of high-value AI solutions in life sciences. This guide explains how to tailor content for diverse stakeho

Explore a 2026 ROI analysis of Quality 4.0 in pharma manufacturing. Learn how technologies like AI & digital twins drive up to 45% cost savings and 40% capacity

Learn what a Case Report Form (CRF) library is and how it improves clinical trials. Covers CDASHIG v2.1, ODM-XML v2.0, Veeva Vault EDC, Medidata Designer AI, and the FDA Dataset-JSON RFC (Apr 2025).

Explore OpenStudyBuilder, the open-source platform for clinical study specifications. Updated for 2026 with ICH M11 adoption, CDISC 360i launch, USDM 4.0, and growing multi-company collaboration.

Updated April 2026 for ServiceNow's Yokohama and Zurich releases, the Moveworks acquisition, and new FDA/EMA AI guidance. A guide to ServiceNow use cases in healthcare and life sciences – ITSM for clinical workflows, patient experience, HIPAA/GxP compliance, EMR integration, and agentic AI.

An in-depth analysis of OpenAI's GPT-5.2 breakthrough on ARC-AGI-2 (54% in Dec 2025), the rapid progress to 98% by April 2026, and the launch of ARC-AGI-3 that humbled all frontier models back to near-zero.

Explore the modern biotech lab's evolution in 2026. Learn how automation, AI, self-driving labs, and digital tools are transforming research, from high-throughput screening to autonomous discovery platforms.

An analysis of the Agentic AI Foundation (AAIF) by the Linux Foundation. Updated April 2026 with 170+ members, MCP's 110M+ monthly downloads, A2A v1.0, and enterprise adoption stats.

Explore document automation for pharma in 2026. Learn how template-driven assembly, structured content, and agentic AI can cut CSR authoring time by 40-90%, with real-world case studies from Merck, Narrativa, and more

Learn how Retrieval-Augmented Generation (RAG) connects ELN, LIMS, and institutional knowledge for drug discovery. Updated for 2026 with FDA/EMA AI guidelines, Benchling AI agents, GraphRAG, and latest market data.

Learn how machine learning (ML) and AI are transforming pharmaceutical CMC process optimization in 2026. Covers applications, digital twins, regulatory updates (FDA/EMA 2026 AI principles, FRAME initiative), and real-world case studies.

Learn how generative AI and LLMs assist in drafting pharma IND & CTA submissions. This guide covers benefits, risks, GxP compliance, FDA/EMA 2026 joint AI principles, and tools like AutoIND and Narrativa.
© 2026 IntuitionLabs. All rights reserved.