
An educational guide to QALYs and ICERs, the core metrics in health economics. Learn how they measure value and guide cost-effectiveness decisions. Updated April 2026 with NICE's new £25k–£35k/QALY threshold.

An educational guide to QALYs and ICERs, the core metrics in health economics. Learn how they measure value and guide cost-effectiveness decisions. Updated April 2026 with NICE's new £25k–£35k/QALY threshold.

An in-depth comparison of ICER (US) vs NICE (UK). Explore their methods, QALY thresholds, and how they assess drug value to influence global pricing floors.

Learn what must be included in a pharma quality agreement. This 2026-updated guide covers key clauses, sponsor vs. CDMO responsibilities, and GMP regulatory requirements including FDA 2016 guidance, EU GMP Annex 1 (2023), ICH Q9(R1), and DSCSA.

Learn the fundamentals of pharmaceutical stability programs. Covers study design, execution, and data trending per the new consolidated ICH Q1 (2024) and FDA guidelines.

Understand analytical method validation with this deep dive into ICH Q2(R2). Explore validation parameters, documentation, and the new QbD lifecycle approach wi

Lifecycle approach to biotech process validation: PPQ and CPV planning, execution, and monitoring under FDA, EMA Annex 1/15, ICH Q9(R1), Q12, and Q13 guidance.

An educational guide to generic drug patent challenges. Learn about the Hatch-Waxman Act, the ANDA pathway, Paragraph IV certifications, the FDA Orange Book, FTC delisting actions, and the 2024 Teva v. Amneal Federal Circuit ruling.

Learn how pharma companies compliantly fund physician education via unrestricted grants. Explore ACCME standards, the Sunshine Act, and firewalls for ethical CM

Learn how Process Analytical Technology (PAT) uses sensors like NIR & Raman for real-time reaction monitoring in pharmaceutical manufacturing to improve quality

Learn how Quality by Design (QbD) and IT systems are shifting pharma from end-product testing to real-time quality assurance with PAT and data analytics.

Updated April 2026: Million-dollar gene therapy pricing analysis covering Casgevy, Lyfgenia, Lenmeldy, Elevidys (with 2025 safety update), the Roctavian and Beqvez market withdrawals, and CMS's 2025 Cell and Gene Therapy Access Model. Compares one-time cure costs to lifetime chronic care using value-based pricing, ICER QALY thresholds, and outcomes-based contracts.

Analysis of how Large Language Models (LLMs) automate clinical evidence collection for HTA economic dossiers. Covers systematic reviews, data extraction, and ch

Learn how patient quality of life (QoL) data is quantified into Quality-Adjusted Life Years (QALYs) to perform cost-effectiveness analysis for specialty drugs.

How real-world data (RWD) and evidence (RWE) validate clinical trial assumptions in post-market economic models. Updated April 2026 with EU JCA, DARWIN EU, FDA finalized RWE guidance, ICH M14 and EHDS developments.

Learn how health economic data like cost-effectiveness analysis is used to secure drug formulary placement and reimbursement from payers and HTA bodies.

Learn the integrated workflow for manufacturing deviations, CAPA, and change control in GMP. This guide covers regulatory requirements and common inspection pit
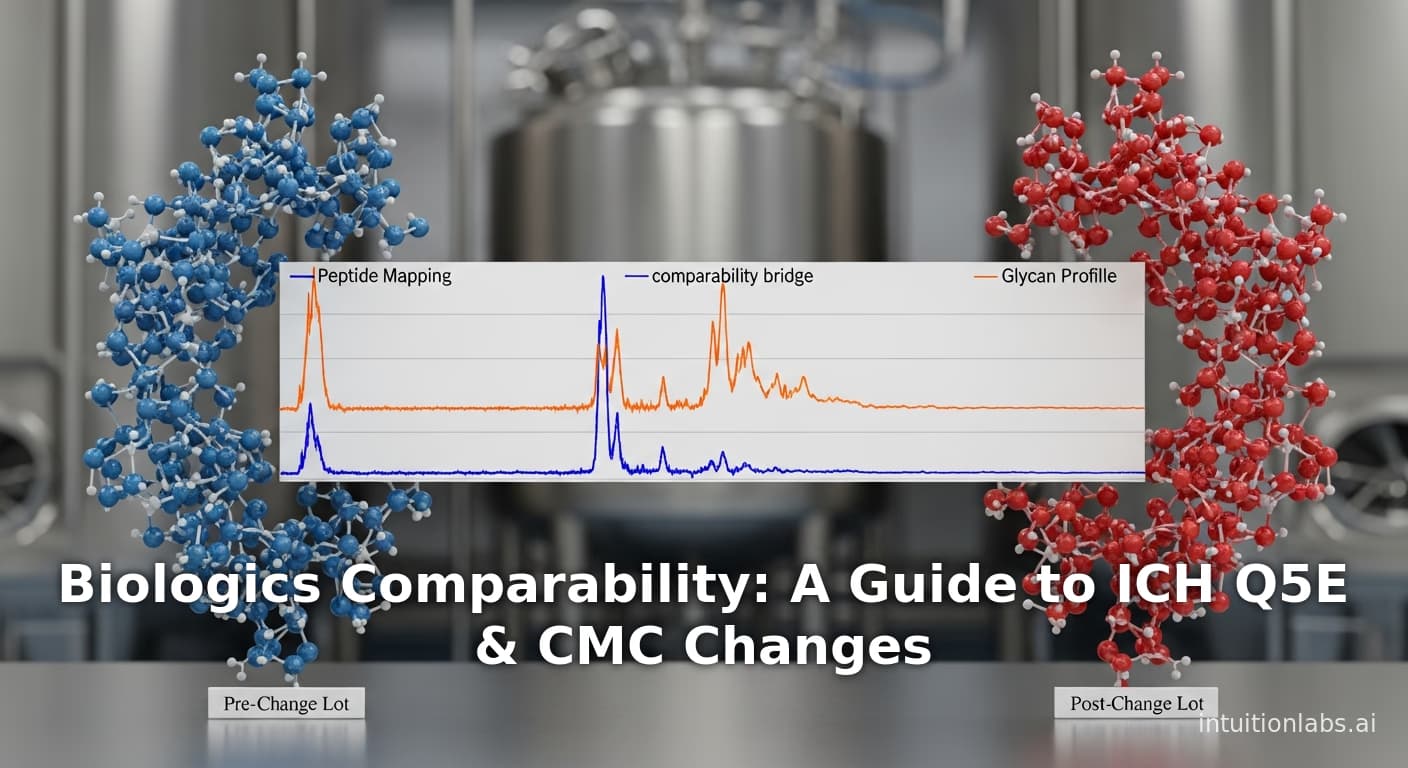
An educational guide to biologics comparability studies. Learn how to prove "no meaningful change" after manufacturing process changes, based on ICH Q5E and FDA

Learn how Integrated Business Planning (IBP) and S&OP help pharma supply chains align demand, capacity, inventory, and constraints. Updated April 2026 with GLP-1 supply lessons, FDA RMP guidance, biosimilar wave 2, and AI in IBP.

Learn how MES and Electronic Batch Records (EBR) improve pharma manufacturing. This guide explains benefits for GMP compliance, data integrity, and review-by-ex

Learn the key differences between US DSCSA and EU FMD for pharmaceutical serialization. This guide covers compliance requirements for ops teams, including data

A proven framework for CDMO selection in pharma and biotech, with scorecards, due diligence checklists, and governance KPIs — updated April 2026 with BIOSECURE Act, Catalent/Novo deal, and EU GMP Annex 1 impacts.

Learn the critical steps for successful pharmaceutical technology transfer. This guide covers the R&D to CDMO process, key documents, and how to avoid scale-up

Learn to validate AI/ML systems in GxP manufacturing using the GAMP 5 Appendix D11 framework. Explore key considerations for data integrity, risk, and model dri

Learn to deploy private LLMs in pharma. This guide covers compliant architecture, including on-premise & cloud, and navigating HIPAA, GxP, and FDA regulations.
© 2026 IntuitionLabs. All rights reserved.