
Explore the top MCP servers for biotech. Learn how the Model Context Protocol connects AI agents and LLMs to critical databases for genomics and drug discovery.

Explore the top MCP servers for biotech. Learn how the Model Context Protocol connects AI agents and LLMs to critical databases for genomics and drug discovery.

Learn the key differences between Anthropic's Claude Skills and the Model Context Protocol (MCP). This guide explains their architecture and use cases.

Explore how AI in remote patient monitoring (RPM) improves clinical outcomes. Updated for 2026 with FDA PCCP guidance, Apple Watch hypertension clearance, CMS reimbursement changes, and market projections.

Learn about FutureHouse, the nonprofit AI research lab. This guide covers its platform, AI agents (Crow, Falcon, Owl, Phoenix, Finch), the Robin discovery system, ether0 reasoning model, and Edison Scientific spinout.

An in-depth analysis of the 2025 Computer System Validation (CSV) job market. Learn about validation specialist salaries, key skills, and career trends.

An in-depth analysis of 1,000+ medical device companies in the SF Bay Area, covering the medtech ecosystem, key players like Intuitive Surgical ($10B+ revenue), Penumbra ($14.5B acquisition), and CeriBell's IPO, plus VC funding and market trends through 2026.

Updated guide to Sora 2, OpenAI's AI video model and TikTok-style app. Covers the Disney $1B deal, pricing changes, Android launch, download decline, and competition from Google Veo 3.

An educational analysis of California's AB 1415 and SB 351. Learn how these laws impact private equity and VC in healthcare, including OHCA review and new rules

Learn about Humanity's Last Exam (HLE), the Nature-published AI benchmark testing true LLM reasoning with 2,500 expert-level questions. Updated with 2026 leaderboard scores from GPT-5, Claude Opus, and Gemini 3.
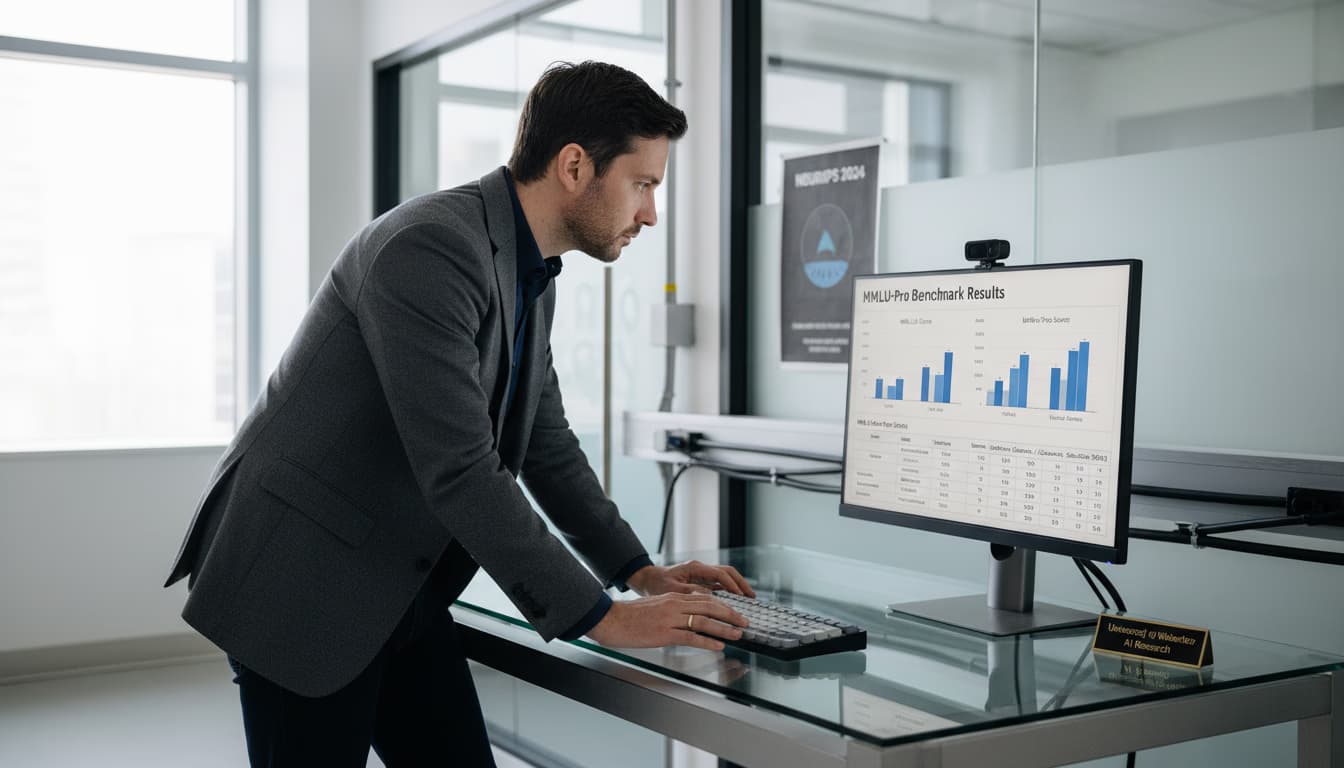
Learn about MMLU-Pro, the advanced AI benchmark designed to overcome MMLU's limitations. This guide explains its design, dataset, and impact on LLM evaluation.

NVIDIA H100 GPU rental rates from $1.49/hr (Vast.ai) to $6.98/hr (Azure). Compare AWS, GCP, Lambda, RunPod, CoreWeave and more.

Updated 2026 comparison of NVIDIA data center GPUs: Blackwell Ultra B300, B200, GB200 NVL72, H100, H200, A100 & L40S — specs, FLOPS, NVLink, cloud availability & Vera Rubin roadmap.

Explore the exponential rise in AI compute demand in biotech. This 2025 report analyzes key statistics, infrastructure needs, and trends in drug discovery and g

Explore the AIME 2025 benchmark, a key test for AI mathematical reasoning. See how models like GPT-5 score over 94% and compare LLM performance on Olympiad-leve

Learn why DeepSeek's AI inference is up to 50x cheaper than competitors. This analysis covers its Mixture-of-Experts (MoE) architecture and pricing strategy.
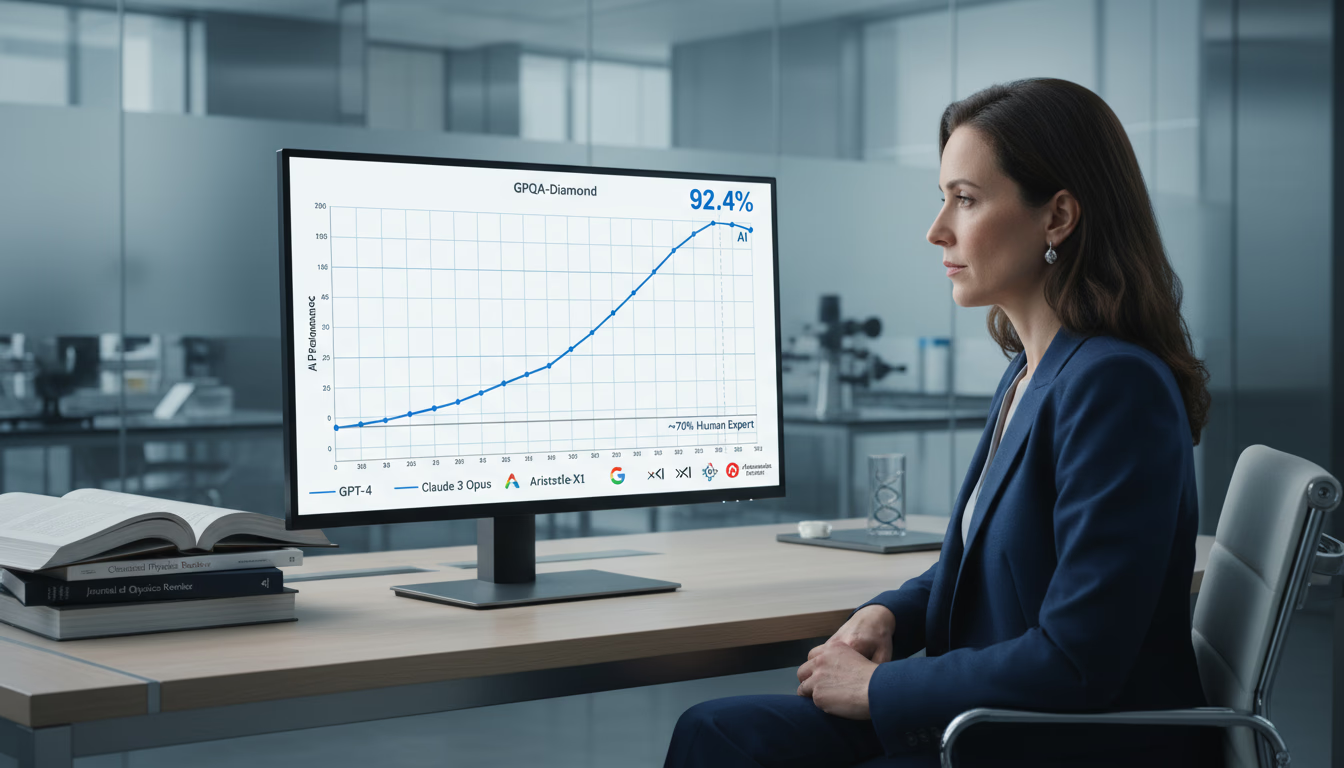
GPQA-Diamond scores updated through 2026: Gemini 3.1 Pro (94.1%), GPT-5.2, Claude Opus 4.6, Aristotle-X1, and more. See which AI models beat PhD experts on 198 graduate-level science questions.

A technical guide to building a HIPAA-compliant OCR pipeline for healthcare. Learn key security controls, PHI handling, encryption, and cloud architecture.

An educational guide to HPC in life sciences. We review top lab IT specialists and solutions for genomics, drug discovery, and bioinformatics data analysis.

Updated 2026: Learn what NVIDIA BioNeMo is and how it accelerates drug discovery. This guide explains its components, latest models (RNAPro, ReaSyn v2), the $1B Lilly partnership, and deployment for generative AI in biopharma.

Learn the strict physical requirements for deploying NVIDIA HGX platforms from Hopper to Blackwell. Covers power (10-140 kW/rack), liquid cooling, rack design, and infrastructure for DGX H100, B200, and GB200 NVL72 systems.

Analyze the leading data center providers for private AI solutions in 2025-2026. This guide compares on-prem and hybrid infrastructure from AWS, Azure, HPE, Dell, Cisco, VMware/Broadcom, Equinix, and NVIDIA, including Blackwell GPUs and sovereign cloud trends

An in-depth analysis of ChatGPT Atlas, OpenAI's AI browser — updated March 2026 with latest feature updates, 900M+ ChatGPT users, Google Chrome's Gemini response, and the evolving AI browser war.

Get a complete 2025 analysis of Clinical Trial Management System (CTMS) software. This guide compares top vendors, core features, and eClinical market trends.

An analysis of how Docusign (formerly DocuSign) is used for electronic signatures in pharma and life sciences. Learn how it can meet FDA 21 CFR Part 11 compliance with its Life Sciences modules and IAM platform, updated for 2026.
© 2026 IntuitionLabs. All rights reserved.