Veeva Services Overview
Tailored consulting and integrations that unlock the full potential of Veeva CRM.
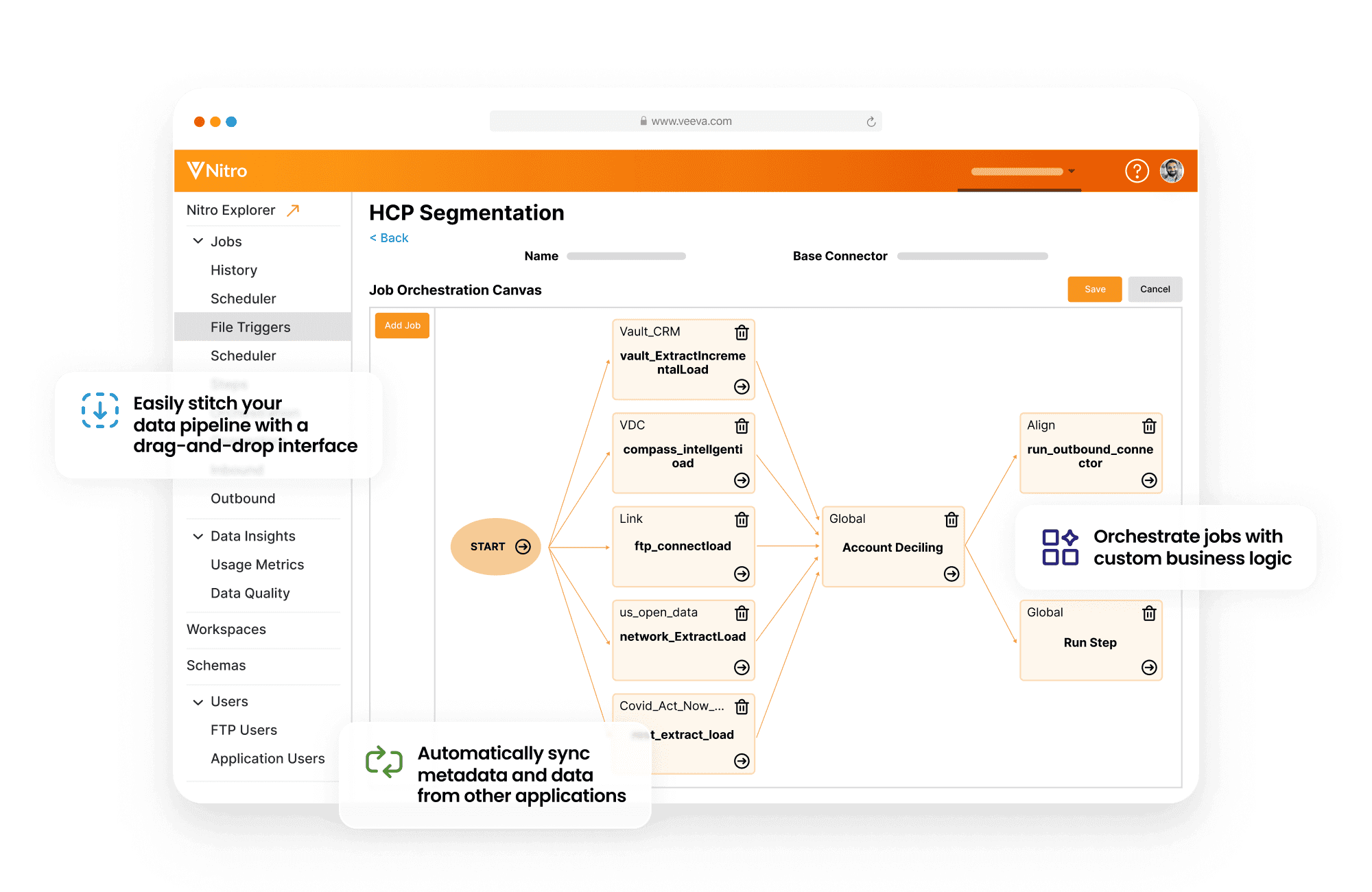
Life sciences teams run on data, but making Veeva fit your exact workflows is rarely straightforward. At IntuitionLabs, we help clinical, regulatory, and commercial teams streamline their CRM experience. From custom dashboards to integrated analytics, our solutions are built to improve clarity, ensure compliance, and strengthen adoption across the organization.

Our Approach
IntuitionLabs is an X-Pages Partner in the Veeva Partner Program
Every project is an opportunity to align strategy with execution. At IntuitionLabs, we draw on deep domain expertise to deliver solutions that are tailored, compliant, and future-proof.
Deep Life Sciences Expertise
AI-First Enhancements
Compliance by Design
Seamless Integration
Training & Support
Frequently Asked Questions
Transform the way your
organization uses Veeva?
With IntuitionLabs, you get trusted expertise, compliance by design, and solutions that scale with you.
Schedule your Veeva Assessment